Articles
- Page Path
- HOME > J Liver Cancer > Volume 22(1); 2022 > Article
-
Case Report
Multidisciplinary treatment with immune checkpoint inhibitors for advanced stage hepatocellular carcinoma -
Ahlim Lee1,2
 , Jaejun Lee1,2
, Jaejun Lee1,2 , Hyun Yang1,2
, Hyun Yang1,2 , Soo-Yoon Sung3
, Soo-Yoon Sung3 , Chang Ho Jeon4
, Chang Ho Jeon4 , Su Ho Kim4
, Su Ho Kim4 , Moon Hyung Choi4
, Moon Hyung Choi4 , Young Joon Lee4
, Young Joon Lee4 , Ho Jong Chun5
, Ho Jong Chun5 , Si Hyun Bae1,2
, Si Hyun Bae1,2
-
Journal of Liver Cancer 2022;22(1):75-83.
DOI: https://doi.org/10.17998/jlc.2022.03.04
Published online: March 18, 2022
1Division of Gastroenterology and Hepatology, Department of Internal Medicine, Eunpyeong St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
2The Catholic University Liver Research Center, College of Medicine, The Catholic University of Korea, Seoul, Korea
3Department of Radiation Oncology, The Catholic University of Korea, Seoul, Korea
4Department of Radiology, Eunpyeong St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
5Department of Radiology, Seoul St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
-
Corresponding author: Si Hyun Bae, Division of Gastroenterology and Hepatology, Department of Internal Medicine, Eunpyeong St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, 1021 Tongil-ro, Eunpyeong-gu, Seoul 03312, Korea,
Tel. +82-2-2030-4312, Fax. +82-2-2030-2521, E-mail: baesh@catholic.ac.kr
Copyright © 2022 by The Korean Liver Cancer Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 4,462 Views
- 98 Downloads
- 4 Citations
Abstract
- Hepatocellular carcinoma (HCC) is a cytotoxic chemotherapy-resistant tumor and most HCCs arise in a background of liver cirrhosis of various causes. Although the IMBrave150 trial showed remarkable advancements in the treatment of unresectable HCC with atezolizumab plus bevacizumab (AteBeva), therapeutic outcomes were unsatisfactory in more than half of the patients. Accordingly, many ongoing trials combine conventional modalities with new drugs such as immune checkpoint inhibitors for better treatment outcomes, and they are expected to benefit patients with limited responses to conventional treatment. Here, two patients with advanced stage HCC with preserved liver function and good performance status showed partial response after treatment with combination or sequential therapy of AteBeva, hepatic arterial infusion chemotherapy, radiation therapy, and transarterial chemoembolization. These findings indicate the efficacy of multidisciplinary treatment against advanced HCC. Additional studies are required to establish optimal treatment strategies.
- Hepatocellular carcinoma (HCC) is a common malignant type of cancer and a leading cause of cancer-related death.1 Because HCC is a heterogeneous disease of multiple etiologies and most HCCs are a consequence of cirrhosis, which impairs liver function, it is challenging to develop standardized therapeutic plans.2–4
- Many ongoing studies examine various treatment regimens for advanced stage HCC, because most HCCs are diagnosed at an advanced stage and are unresectable; therefore, treatment options are limited to palliative care.3,5,6 For better treatment outcomes, many clinical trials have examined the efficacy of multidisciplinary treatments and the related mechanisms.2,5,7–11
- Previous reports have shown that hepatic arterial infusion chemotherapy (HAIC) improved the survival rate in patients with intrahepatic tumors, even in patients with extrahepatic metastases or portal vein tumor thrombosis (PVTT).12,13 Additionally, the recent IMBrave150 trial proved that the overall survival and median progression-free survival (PFS) duration of patients submitted to combination therapy with atezolizumab plus bevacizumab (AteBeva) were superior to those of patients treated with sorafenib.
- Here, we report two cases of advanced stage HCC with PVTT or multiple lung metastases, which were treated with combination or sequential therapy of AteBeva based on HAIC and showed partial response (PR). These cases are reported according to the CARE guidelines available at https://www.care-statement.org/.
INTRODUCTION
- The first case was a 66-year-old man who visited our clinic complaining of abdominal distension and weight loss of 7 kg over the previous 3 months. He had hypertension and chronic hepatitis B infection and was being treated with 300 mg tenofovir disoproxil fumarate once daily for several years. On physical examination, he presented with tenderness in the right upper quadrant.
- Complete blood count showed hemoglobin level of 7.0 g/dL, leukocyte count of 6,500/μL, and platelet count of 153,000/μL. Laboratory examinations showed serum aspartate aminotransferase (AST) 44 IU/L, alanine aminotransferase (ALT) 32 IU/L, alkaline phosphatase (ALP) 216 IU/L, gamma-glutamyltransferase (GGT) 49 U/L, total bilirubin level 0.62 mg/dL, total protein 7.60 g/dL, and albumin 2.81 g/dL. The international normalized ratio (INR) of prothrombin time was 1.45. Hepatitis B surface antigen and envelope antibody were positive, and the hepatitis B virus DNA level was 477 IU/mL.
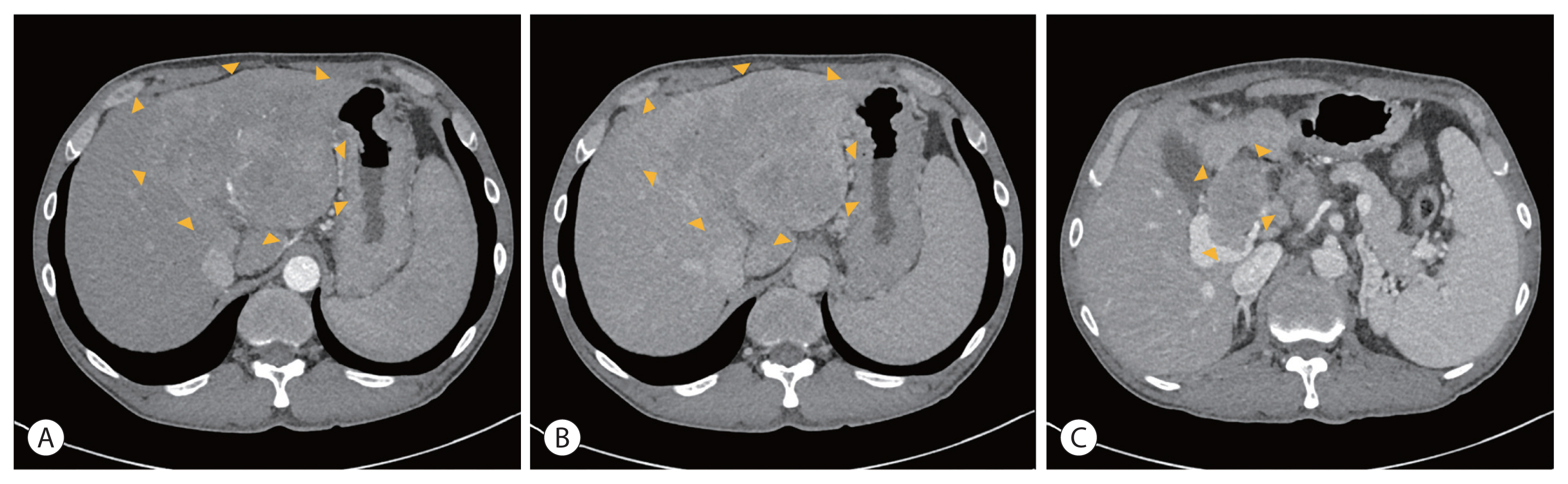
- Liver dynamic computed tomography (CT) showed that approximately 12.8 cm of the heterogeneously enhanced mass occupied the left lobe in the arterial phase, with washout in the delayed phase. Additionally, a left portal vein thrombus extending to the main portal vein was observed in the portal phase (Fig. 1). Initial alpha-fetoprotein (AFP) was 538.25 ng/mL (normal range, ≤9 mg/dL) and prothrombin-induced by vitamin K absence or antagonist-II (PIVKA-II) was 146.54 mAU/mL (normal range, ≤40 mAU/mL).
- Based on clinical and imaging findings, the hepatic mass was diagnosed as HCC. Chest CT did not show any evidence of lung metastasis. Child-Pugh score of 6, class A, and Eastern Cooperative Oncology Group (ECOG) score of 0. The patient was classified according to the modified Union for International Cancer Control (mUICC) as stage IVa (T4N0M0) and as advanced stage (stage C) according to the Barcelona Clinic Liver Cancer (BCLC) staging system. We performed esophagogastroduodenoscopy before starting HCC treatment and found esophageal varices of form 0 at the lower esophagus and no gastric varix.
- Systemic treatment was recommended as the first-line treatment of mUICC IVa and BCLC C according to the Korea Practice Guidelines for Patients with HCC from the Korean Liver Cancer Association-National Cancer Center 2018 and BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update.14,15 Therefore, we chose a treatment regimen including AteBeva, which is a first-line systemic treatment. Simultaneously, we performed HAIC based on 5-fluorouracil (5-FU) and cisplatin, because previous reports have shown that HAIC improved the survival rate in patients with intrahepatic tumors, even in those with extrahepatic metastases or PVTT.12,13 In addition, another report has shown that the outcome of advanced stage HCC was better after treatment with HAIC with anti-programmed cell death protein-1 immunotherapy than HAIC alone in terms of overall survival, PFS, disease control rate, and intrahepatic response.11 In this case, atezolizumab (Ticentriq®, 1,200 mg per dose; Roche International LLC, Basel, Switzerland) and bevacizumab (Avastin®, 15 mg/kg per dose; Genentech, South San Francisco, CA, USA) were administered 1 day every 3 weeks. For HAIC, 500 mg/m2 5-FU for 3 days and 60 mg/m2 cisplatin for 1 day were administered. HAIC was performed at 3-week intervals on the day after AteBeva administration. Response was evaluated after every three cycles of AteBeva with HAIC treatment.
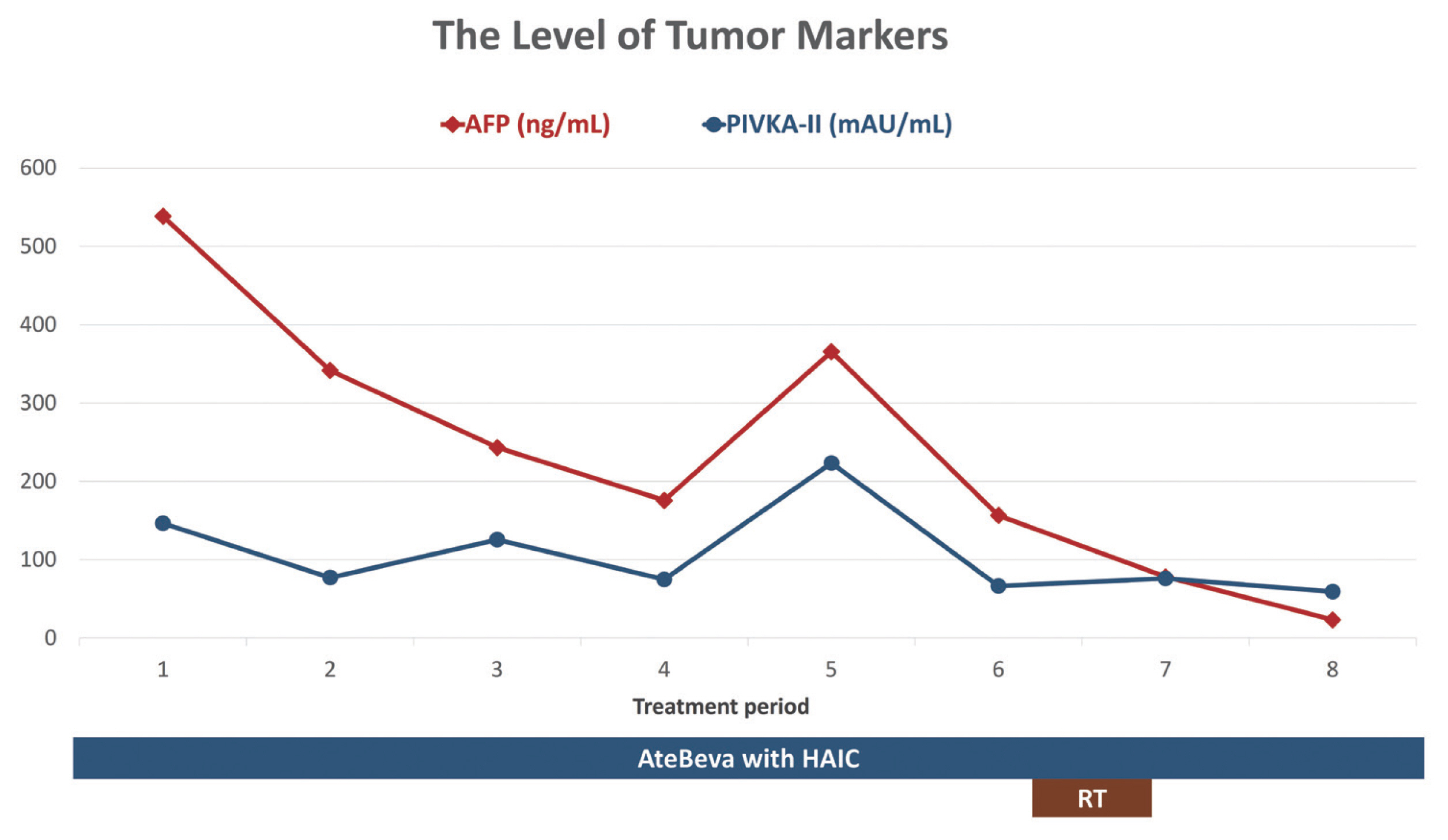
- After the 4th cycle of AteBeva and HAIC, follow-up liver dynamic CT showed decreased size of the intrahepatic mass and left PVTT but an extension of PVTT to the right portal vein, and increased AFP and PIVKA-II levels. Therefore, radiation therapy (RT) was provided to the remaining hepatic mass and PVTT. A total dose of 45 Gy was administered 10 times at 450 cGy each, continuing with AteBeva and HAIC at 3-week intervals.
- After the 8th treatment cycle with AteBeva and HAIC with RT, follow-up gadoxetic acid-enhanced liver magnetic resonance imaging (MRI) showed little interval change in the size of the intrahepatic mass and the size and extent of PVTT with no definite enhancing viable area compared with the previous liver image (Fig. 2). The levels of tumor markers decreased (Fig. 3). Treatment response was determined as PR based on the modified Response Evaluation Criteria in Solid Tumors, version 1.1 (mRECIST 1.1) for HCC.
- The second case involved a 54-year-old man who visited our clinic for further evaluation of a hepatic mass and impaired liver function test results after a regular check-up. He had been taking medication for hypertension and dyslipidemia for several years. He was a 45 pack-year current smoker who consumed approximately 600 g of alcohol every week. At the time of presentation, the patient did not experience any abdominal discomfort.
- Complete blood count showed hemoglobin level of 9.2 g/dL, leukocyte count of 9,800/μL, and platelet count of 252,000/μL. Laboratory examinations showed serum AST 88 IU/L, ALT 50 IU/L, ALP 78 IU/L, GGT 371 U/L, total bilirubin level 1.01 mg/dL, total protein 7.60 g/dL, albumin 3.62 g/dL, and INR of prothrombin time was 1.31. Hepatitis B surface antigen, core antibody, and hepatitis C viral antibody tests were negative.
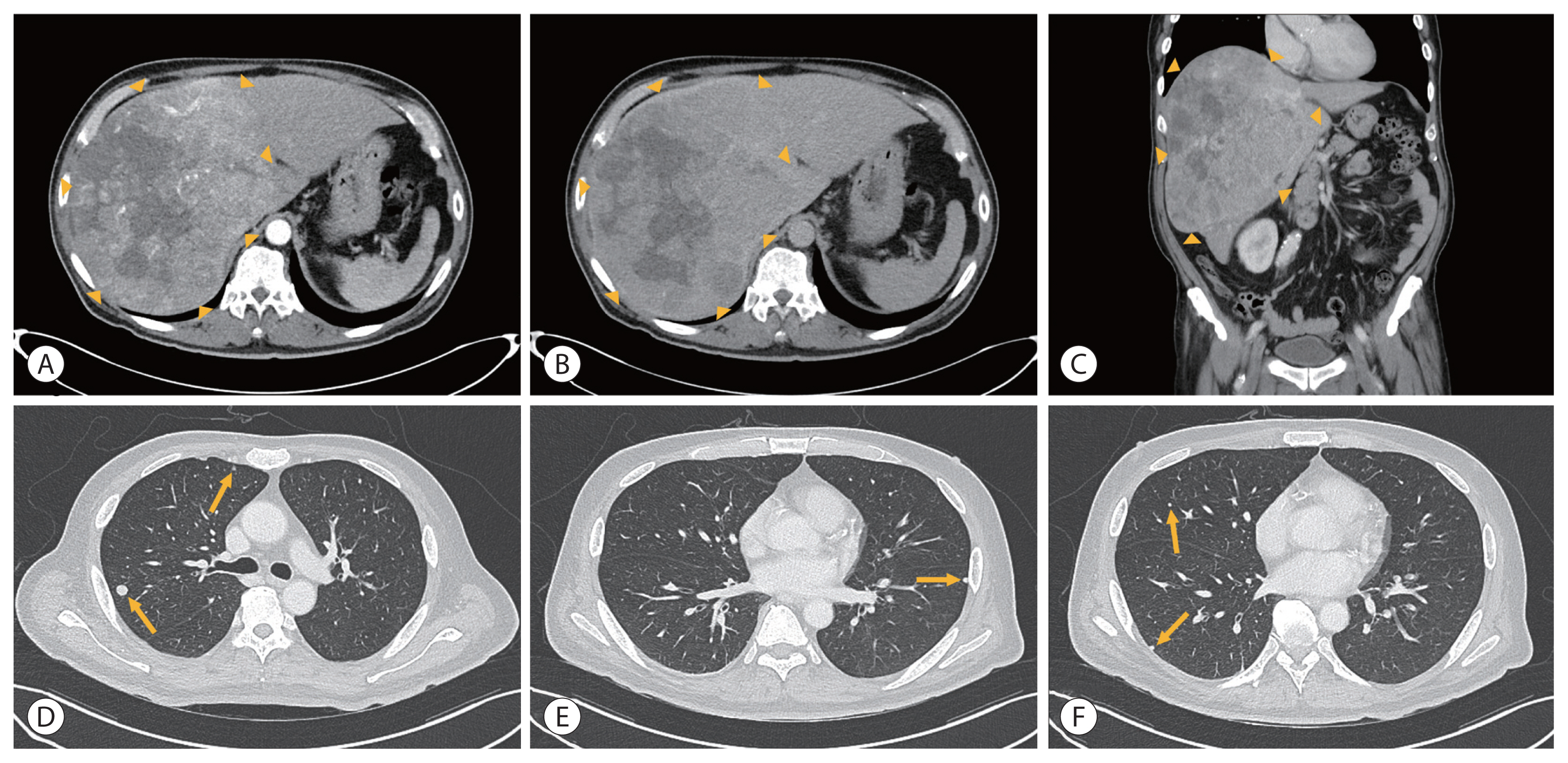
- Liver dynamic CT showed that approximately 23.5×13.7 cm of the heterogeneously enhanced mass occupied the right lobe in the arterial phase with washout in the delayed phase and no evidence of PVTT (Fig. 4).
- Based on the history of alcohol intake and the clinical and imaging findings, the hepatic mass was diagnosed as HCC. A CT scan of the chest showed multiple metastases in both lung fields. Child-Pugh score of 5, class A and ECOG score of 0. This patient was diagnosed with mUICC stage IVb, T4N0M1, and BCLC stage C. Initial AFP was 6,528 ng/mL and PIVKA-II was 100,000 mAU/mL. Esophagogastroduodenoscopy before starting HCC treatment showed no esophageal or gastric varices.
- Previous studies have found that controllability of intrahepatic tumors and hepatic reserves were significant predictors of survival in patients with advanced HCC and extrahepatic metastases, emphasizing the importance of therapeutic approaches to control intrahepatic tumors.16,17 We chose a treatment option with transarterial chemoembolization (TACE), because of the high tumor burden of the intrahepatic mass of the patient. In addition, we added HAIC, because previous reports showed good treatment efficacy of HAIC against advanced stage HCC with extrahepatic metastases and proved that the overall response rate of HAIC with TACE was superior to that of TACE alone.12,13,18 For conventional TACE, a mixture of 10 mL iodized oil (Lipiodol; Laboratoire Guerbet, Aulnay-Sous-Bois, France) and 50 mg doxorubicin was infused into the selected feeding arteries. Embolization was performed using absorbable gelatin sponge particles (Gelfoam; Upjohn, Kalamazoo, MI, USA) cut into 1 mm2 pieces. For HAIC, 500 mg/m2 5-FU for 3 days and 60 mg/m2 cisplatin for 1 day were administered at 3-week intervals.
- After the 3rd cycle of TACE and the 5th cycle of HAIC, follow-up liver dynamic CT showed decreased, although remaining, arterial enhancement in the treated HCC in the right lobe, which was considered as the viable portion of the existing tumor. Additionally, follow-up chest CT revealed increased size and number of multiple lung nodules, which corresponded to disease progression according to mRECIST 1.1 (Fig. 5). We changed the therapeutic plan to first-line systemic therapy using AteBeva. A fixed dose of 1,200 mg atezolizumab and 15 mg/kg of bevacizumab was administered at 3-week intervals. Response was evaluated after every three cycles of AteBeva.
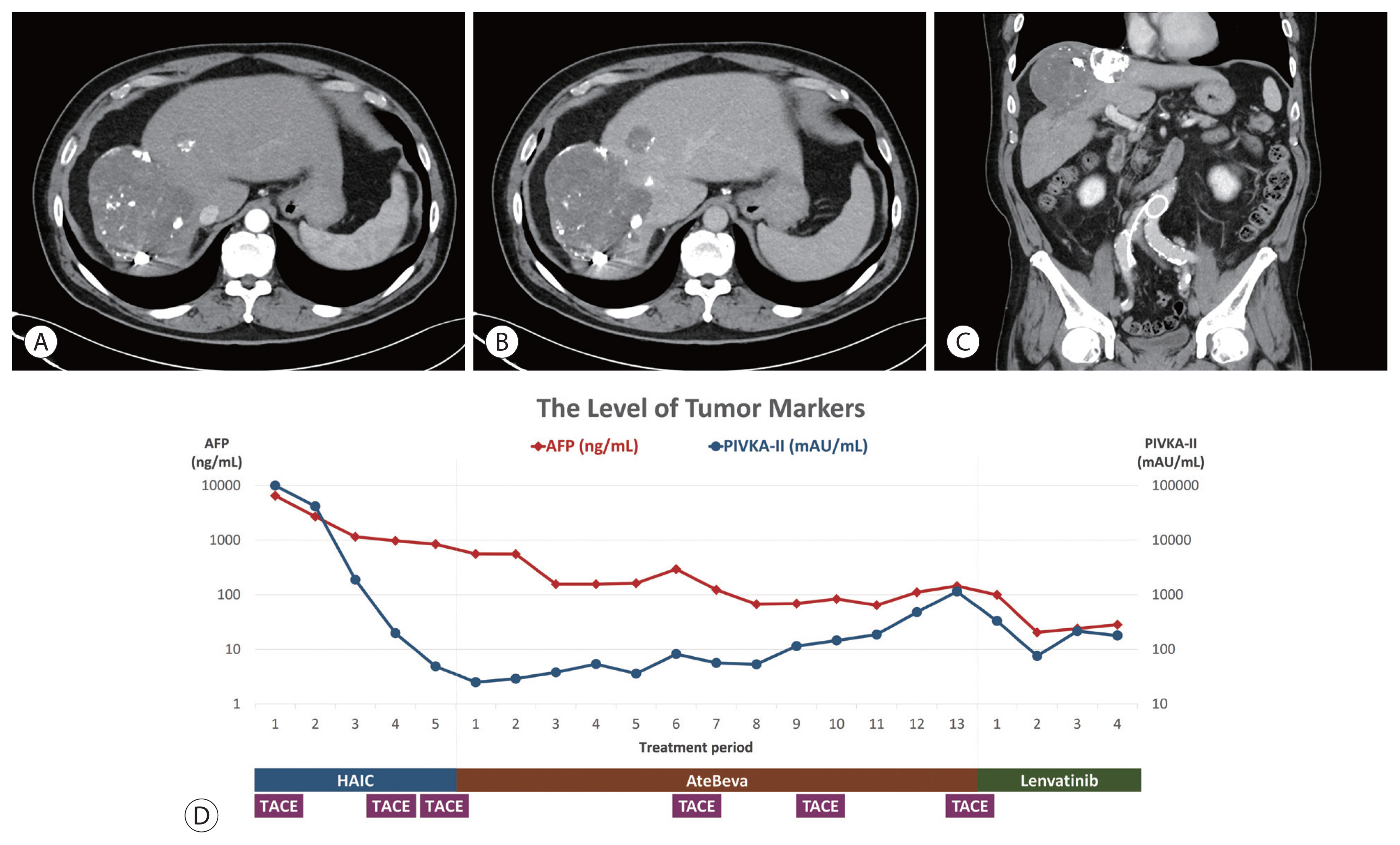
- After three doses of AteBeva, multiple lung metastases decreased according to follow-up chest CT, but two new ovoid masses less than 1.9 cm at the anterior portion of the right hepatic dome with arterial enhancement and early washout in the portal phase were observed on follow-up gadoxetic acid-enhanced liver MRI. We performed TACE three times while maintaining AteBeva, and the follow-up liver CT showed no definite viable portion on treated HCCs in the arterial phase and obtained radiological complete response for the intrahepatic mass.
- However, after an additional 10 doses of AteBeva, follow-up chest CT showed increased size and number of nodules in both lungs, indicative of disease progression. The treatment regimen was changed to lenvatinib (Lenvima®, 12 mg per day; Eisai Co., Ltd., Tokyo, Japan), which is a tyrosine kinase inhibitor (TKI). After 3 months of treatment with the new medication, follow-up chest CT showed little interval change in the size and number of lung nodules, and follow-up liver dynamic CT showed multiple lipiodol uptakes in treated HCCs without definite evidence of viable arterial enhancement. Total treatment response was defined as PR, based on mRECIST 1.1 (Fig. 6).
CASE REPORT
- HCC is the most common malignant tumor of the liver with a high mortality rate because most HCCs are diagnosed at an advanced stage.10 Therefore, advanced stage HCCs have a poor prognosis and, treatment is mostly palliative.5 Recent studies have found that immune checkpoint inhibitors (ICIs) are a promising therapeutic option for advanced stage HCC and have focused on the potential combination of ICIs with tumor microenvironment-modifying agents.9 The IMbrave150 trial showed that the survival rate of advanced stage HCC patients was higher after treatment with atezolizumab - an antibody against programmed cell death ligand 1 (PD-L1) - plus bevacizumab, an anti-vascular endothelial growth factor (VEGF) antibody, than with sorafenib.9 However, although anti-PD-L1 plus anti-VEGF improved treatment outcomes, approximately two-thirds of advanced stage HCC patients still do not benefit from treatment.5 Thus, a large number of ongoing studies aim to improve the treatment outcome with ICIs alone or in combination with systemic or loco-regional therapies.5
- Our first case was a patient with stage IVa (T4N0M0) (according to mUICC), or advanced stage (according to BCLC) HCC, who had chronic hepatitis B infection. We chose a combination therapy of AteBeva every 3 weeks followed by HAIC based on 5-FU with cisplatin because our previous study showed that a pretreatment tumor-to-liver apparent diffusion coefficient (ADC) ratio of <0.741 on MRI was a predictor of an objective response to HAIC.6 Since the pretreatment ADC ratio on MRI was 0.592, we expected a good response to HAIC. However, after the 4th cycle of AteBeva with HAIC, liver dynamic CT revealed a decreased intrahepatic mass but an extension of the right PVTT. We added RT to the remaining intrahepatic mass and PVTT because previous studies found that RT promotes localized tumor killing and potentiates the action of ICIs by modifying the tumor microenvironment.9 After the 8th cycle of treatment, we obtained PR on intrahepatic mass and stable disease on PVTT; the total response was determined as PR.
- The second case was an HCC patient who had been consuming alcohol for several years. His mUICC stage was IVb (T4N0M1) and BCLC stage was C. We chose a treatment option with TACE and HAIC to control the intrahepatic tumor. The patient’s pretreatment ADC ratio on MRI was 0.643, and a good response to HAIC was expected. However, due to progression of lung metastases after the 3rd cycle of TACE and the 5th cycle of HAIC, we changed the regimen to AteBeva for systemic therapy. This therapy improved metastatic lung lesions, however two intrahepatic viable lesions were soon discovered. We added TACE because it may elicit “immunogenic cell death” by exposing tumor antigens, thus promoting antitumor immunity.7 The combination of ICIs with TACE is expected to enhance the antitumor response without excessive toxicity, based on recent preliminary data from single-arm studies.7 However, because the lung metastases progressed after the 13th AteBeva administration, we changed the treatment regimen to lenvatinib. After 3 months, the total response was PR, with maintained radiological complete response in the intrahepatic lesion and little change in lung metastases. A recent multinational multicenter retrospective study demonstrated the efficacy of lenvatinib on PFS in patients with advanced HCC with progression after AteBeva treatment.19 This report was the first to investigate second-line treatment after progression despite AteBeva treatment in HCC patients; however, the mechanisms involved in lenvatinib treatment remain unclear. Further studies are required to determine the various treatment options and the mechanism of the second-line regimen after AteBeva treatment.
- Multidisciplinary HCC treatment was accompanied by several adverse events. In the first patient, renal function decline and bone marrow suppression occurred as 5-FU and cisplatin-based chemotherapy progressed. Therefore, we reduced the dose of 5-FU and cisplatin to up to 25% of the baseline. The second patient developed hypothyroidism related to atezolizumab administration. Levothyroxine was administered to replace his thyroid function.
- HCC is a complex disease with various etiologies. Impaired liver function due to cirrhosis worsens prognosis and complicates clinical decisions for treatment.2 The efficacy of many new drugs, including ICIs, has been proven for treatment against advanced stage HCC. Combination and sequential therapies with HAIC and ICIs, TKIs, anti-VEGF, and loco-regional treatments such as TACE, transarterial radioembolization, RT, and radiofrequency ablation are currently on the trials and, they are considered to have treatment potentials in highly selected patients.2 Continuous and additional large-scale studies are required to examine treatment outcomes from various combinations and to establish the best treatment strategies. Through these efforts, many patients with advanced HCC may benefit from individualized treatment with various options.
DISCUSSION
-
Conflicts of Interest
The authors have no conflicts of interest to disclose.
-
Ethics Statement
The Institutional Review Board of the Catholic University of Korea, Eunpyeong St. Mary’s hospital waived the requirement for ethics approval and informed consent (IRB No. PC21ZISI0182).
-
Funding Statement
No funding to declare.
-
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed for this case report.
-
Author Contribution
Conceptualization: SHB
Data curation: AL, HY, SHB
Formal analysis: HY, SYS, MHC, YJL, HJC, SHB
Project administration: AL, SHB
Resources: JL, CHJ, SHK, HJC
Writing original draft: AL
Writing review & editing: AL, HY, SHB
Approval of final manuscript: all authors
Article information


- 1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021;71:209−249.ArticlePubMed
- 2. Liu PH, Huo TI, Miksad RA. Hepatocellular carcinoma with portal vein tumor involvement: best management strategies. Semin Liver Dis 2018;38:242−251.ArticlePubMed
- 3. Lurje I, Czigany Z, Bednarsch J, Roderburg C, Isfort P, Neumann UP, et al. Treatment strategies for hepatocellular carcinoma—a multidisciplinary approach. Int J Mol Sci 2019;20:1465. ArticlePubMedPMC
- 4. Yang Z, Abdrakhimov B, Wang S, Xie QF, Zheng SS. Multidisciplinary treatment of advanced hepatocellular carcinoma. Case Rep Gastroenterol 2020;14:98−102.ArticlePubMedPMC
- 5. Pinter M, Jain RK, Duda DG. The current landscape of immune checkpoint blockade in hepatocellular carcinoma: a review. JAMA Oncol 2021;7:113−123.ArticlePubMedPMC
- 6. Sung PS, Choi MH, Yang H, Lee SK, Chun HJ, Jang JW, et al. Diffusion-weighted magnetic resonance imaging in hepatocellular carcinoma as a predictor of a response to cisplatin-based hepatic arterial infusion chemotherapy. Front Oncol 2020;10:600233. ArticlePubMedPMC
- 7. Cheng AL, Hsu C, Chan SL, Choo SP, Kudo M. Challenges of combination therapy with immune checkpoint inhibitors for hepatocellular carcinoma. J Hepatol 2020;72:307−319.ArticlePubMed
- 8. Doycheva I, Thuluvath PJ. Systemic therapy for advanced hepatocellular carcinoma: an update of a rapidly evolving field. J Clin Exp Hepatol 2019;9:588−596.ArticlePubMedPMC
- 9. Lee YH, Tai D, Yip C, Choo SP, Chew V. Combinational immunotherapy for hepatocellular carcinoma: radiotherapy, immune checkpoint blockade and beyond. Front Immunol 2020;11:568759. ArticlePubMedPMC
- 10. Liu BJ, Gao S, Zhu X, Guo JH, Kou FX, Liu SX, et al. Real-world study of hepatic artery infusion chemotherapy combined with anti-PD-1 immunotherapy and tyrosine kinase inhibitors for advanced hepatocellular carcinoma. Immunotherapy 2021;13:1395−1405.ArticlePubMed
- 11. Mei J, Li SH, Li QJ, Sun XQ, Lu LH, Lin WP, et al. Anti-PD-1 immunotherapy improves the efficacy of hepatic artery infusion chemotherapy in advanced hepatocellular carcinoma. J Hepatocell Carcinoma 2021;8:167−176.ArticlePubMedPMC
- 12. Sung PS, Yang K, Bae SH, Oh JS, Chun HJ, Nam HC, et al. Reduction of intrahepatic tumour by hepatic arterial infusion chemotherapy prolongs survival in hepatocellular carcinoma. Anticancer Res 2019;39:3909−3916.ArticlePubMed
- 13. Lin CC, Hung CF, Chen WT, Lin SM. Hepatic arterial infusion chemotherapy for advanced hepatocellular carcinoma with portal vein thrombosis: impact of early response to 4 weeks of treatment. Liver Cancer 2015;4:228−240.ArticlePubMedPMC
- 14. Korean Liver Cancer Association; National Cancer Center. 2018 Korean Liver Cancer Association-National Cancer Center Korea practice guidelines for the management of hepatocellular carcinoma. Gut Liver 2019;13:227−299.ArticlePubMedPMC
- 15. Reig M, Forner A, Rimola J, Ferrer-Fàbrega J, Burrel M, Garcia-Criado Á, et al. BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update. J Hepatol 2022;76:681−693.ArticlePubMed
- 16. Jung SM, Jang JW, You CR, Yoo SH, Kwon JH, Bae SH, et al. Role of intrahepatic tumor control in the prognosis of patients with hepatocellular carcinoma and extrahepatic metastases. J Gastroenterol Hepatol 2012;27:684−689.ArticlePubMed
- 17. Uchino K, Tateishi R, Shiina S, Kanda M, Masuzaki R, Kondo Y, et al. Hepatocellular carcinoma with extrahepatic metastasis: clinical features and prognostic factors. Cancer 2011;117:4475−4483.ArticlePubMed
- 18. Gao S, Zhang PJ, Guo JH, Chen H, Xu HF, Liu P, et al. Chemoembolization alone vs combined chemoembolization and hepatic arterial infusion chemotherapy in inoperable hepatocellular carcinoma patients. World J Gastroenterol 2015;21:10443−10452.ArticlePubMedPMC
- 19. Yoo C, Kim JH, Ryu MH, Park SR, Lee D, Kim KM, et al. Clinical outcomes with multikinase inhibitors after progression on first-line atezolizumab plus bevacizumab in patients with advanced hepatocellular carcinoma: a multinational multicenter retrospective study. Liver Cancer 2021;10:107−114.ArticlePubMedPMC
References
Figure & Data
References
Citations

- Complications of immunotherapy in advanced hepatocellular carcinoma
Young-Gi Song, Jeong-Ju Yoo, Sang Gyune Kim, Young Seok Kim
Journal of Liver Cancer.2024; 24(1): 9. CrossRef - Higher objective responses by hepatic arterial infusion chemotherapy following atezolizumab and bevacizumab failure than when used as initial therapy in hepatocellular carcinoma: a retrospective study
Jae-Sung Yoo, Ji Hoon Kim, Hee Sun Cho, Ji Won Han, Jeong Won Jang, Jong Young Choi, Seung Kew Yoon, Suho Kim, Jung Suk Oh, Ho Jong Chun, Pil Soo Sung
Abdominal Radiology.2024;[Epub] CrossRef - Feasibility of additional radiotherapy in patients with advanced hepatocellular carcinoma treated with atezolizumab plus bevacizumab
Tae Hyun Kim, Bo Hyun Kim, Yu Ri Cho, Young-Hwan Koh, Joong-Won Park
Journal of Liver Cancer.2023; 23(2): 330. CrossRef - Is multidisciplinary treatment effective for hepatocellular carcinoma with portal vein tumor thrombus?
Won Hyeok Choe
Journal of Liver Cancer.2022; 22(1): 1. CrossRef
 PubReader
PubReader ePub Link
ePub Link Download Citation
Download Citation
- Download Citation
- Close
- Related articles
-
- Complications of immunotherapy in advanced hepatocellular carcinoma
- A multidisciplinary approach with immunotherapies for advanced hepatocellular carcinoma
- The role of lenvatinib in the era of immunotherapy of hepatocellular carcinoma
- How to optimize the treatment strategy for advanced-stage hepatocellular carcinoma with macrovascular invasion
- Adult hepatoblastoma: making the challenging distinction from hepatocellular carcinoma

 E-submission
E-submission THE KOREAN LIVER CANCER ASSOCIATION
THE KOREAN LIVER CANCER ASSOCIATION


 Follow JLC on Twitter
Follow JLC on Twitter