Articles
- Page Path
- HOME > J Liver Cancer > Volume 23(2); 2023 > Article
-
Case Report
Hepatic basidiobolomycosis masquerading as cholangiocarcinoma: a case report and literature review -
Roopali Sehrawat1
 , Nalini Bansal1
, Nalini Bansal1 , Ajitabh Srivastava2
, Ajitabh Srivastava2 , Dharmender Malik3
, Dharmender Malik3 , Vivek Vij4
, Vivek Vij4
-
Journal of Liver Cancer 2023;23(2):389-396.
DOI: https://doi.org/10.17998/jlc.2023.06.07
Published online: August 17, 2023
1Department of Pathology, SGT University, Budhera, Gurugram, India
2Department of Liver Transplant Surgery, Aakash Healthcare Super Speciality Hospital, Dwarka, New Delhi, India
3Department of Nuclear Medicine & PET-CT, Fortis Memorial Research Institute (FMRI), Gurugram, India
4Department of Liver Transplant Surgery, Fortis Healthcare, Budhera, Gurugram, India
-
Corresponding author: Nalini Bansal, Department of Pathology, SGT University, Gurgaon-Badli Road Chandu, Budhera, Gurugram 122505, India
Tel. +91-999-037-0677 E-mail: drnalinibansal@gmail.com
© 2023 The Korean Liver Cancer Association.
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 1,247 Views
- 57 Downloads
Abstract
- Basidiobolus ranarum is known to cause subcutaneous mycoses; however, rare cases of hepatic and gastrointestinal involvement by basidiobolomycosis have been reported. Hepatic basidiobolomycosis may be confused with a carcinoma on imaging, and histological examination and fungal culture can help distinguish between these two. We report a rare case of basidiobolomycosis in a 16-year-old male with liver and gastrointestinal involvement.
- Basidiobolomycosis is a rare infection caused by the fungus Basidiobolus ranarum (B. ranarum) belongs to the class Zygomycetes and the family Entomophthoraceae. It is commonly found in warm rural regions of Sub-Saharan Africa, South America, and Asia (Saudi Arabia, Kuwait, Iran, India, Indonesia, and Burma). Basidiobolomycosis mainly affects immunocompetent humans of all age groups, and the infection is usually aggressive and sometimes fatal. The infection often presents with non-specific clinical and radiological features; therefore, it should be considered in the differential diagnosis in endemic regions.1 We report a rare case of basidiobolomycosis in a 16-year-old male with liver and gastrointestinal involvement.
INTRODUCTION
- A 16-year-old male from Iran presented with multiple episodes of vomiting and low oral intake. The patient had a history of seizures and type II diabetes mellitus. On examination, the patient was afebrile; blood pressure was 120/70 mmHg, heart rate was 130/min, respiratory rate was 20/min, oxygen saturation was 98% at room air, and chest was clear bilaterally.
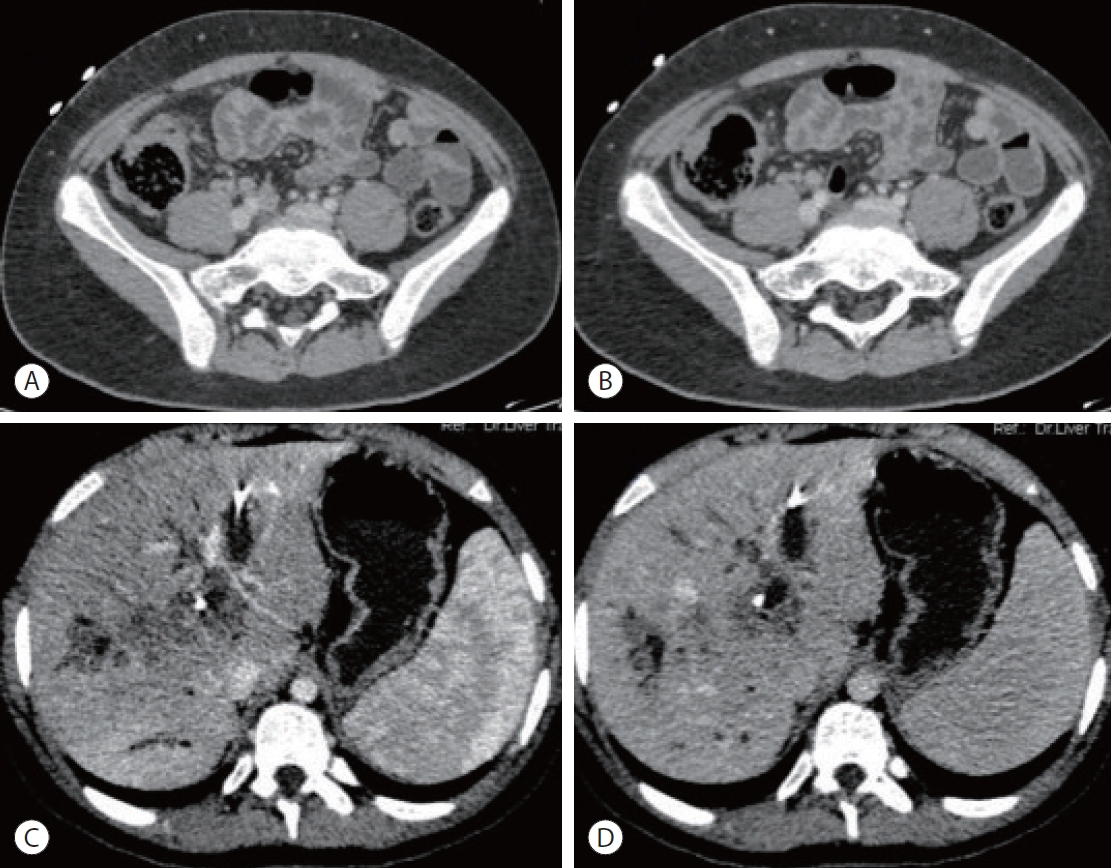
- Abdominal ultrasonography revealed a borderline enlarged liver with a hypoechoic lesion measuring 6.7×6.1 cm in the portal confluence region involving segments 5, 6, and 7. Color Doppler imaging revealed mild color flow within the lesion. Magnetic resonance imaging (MRI) also revealed an enlarged liver (17 cm in size). A lesion with heterogeneous signal intensity showing areas of diffuse restriction measuring 8×7×6.7 cm was seen involving the right lobe of the liver. There was a mass effect on the central intrahepatic biliary tree with consequent non-visualization of the right and left main hepatic ducts, their confluence, and the proximal common hepatic duct. The proximal parts of the intrahepatic biliary radicles were observed within the lesion. Diffusionweighted imaging and the corresponding apparent diffusion coefficient (ADC) images showed areas of diffusion restriction with low ADC values in and around the lesion involving the right hepatic lobe. In pre- and post-contrast mDIXON (T1W) studies, heterogeneous enhancement with a few central non-enhancing areas was observed within the lesion. These findings were suggestive of hilar cholangiocarcinoma, and surgical resection was considered. Coronal post-contrast mDIXON images showed thickening and enhancement of the cecum and ascending colon and heterogeneously enhancing lesions in the liver (Fig. 1). A positron emission tomography-computed tomography (CT) scan showed an enlarged liver measuring 18 cm in the maximum craniocaudal extent. A large, fluorodeoxyglucose (FDG)-avid hypodense lesion was seen in the right lobe of the liver (segments 5-8), measuring approximately 7.0×8.0×6.4 cm. Ulceration with irregular thickening was observed in the cecum, distal ileum and ascending colon, with a maximum mural thickness of 1.5 cm. A few enlarged FDG-avid paracecal lymph nodes were also noted, the largest measuring 1.9 cm along the short axis (Fig. 2).
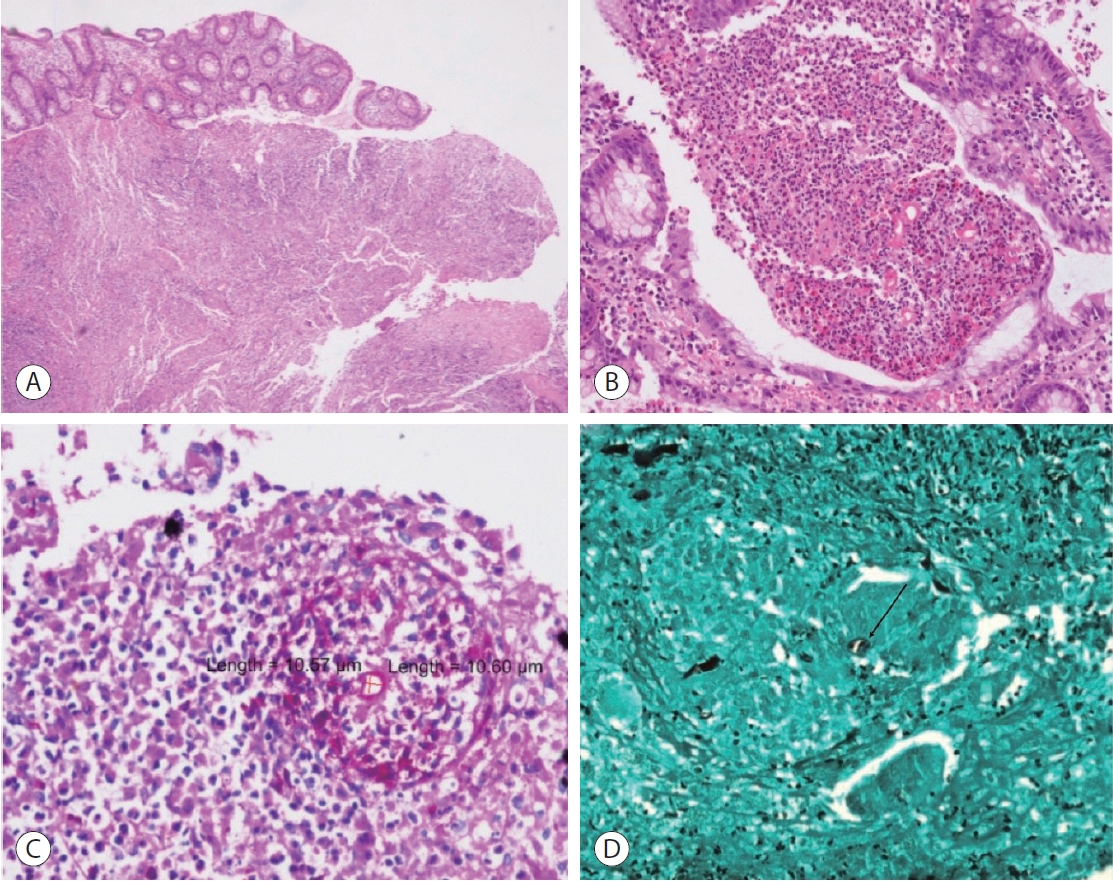
- Percutaneous transhepatic biliary drainage and liver biopsy were performed, and the biopsy specimen was sent for histopathological examination and fungal culture. The biopsy specimen was mostly composed of fibrocollagenous tissue with inflammatory infiltrates and granulomatous reactions. Fungal spores were observed within some giant cells (Fig. 3).
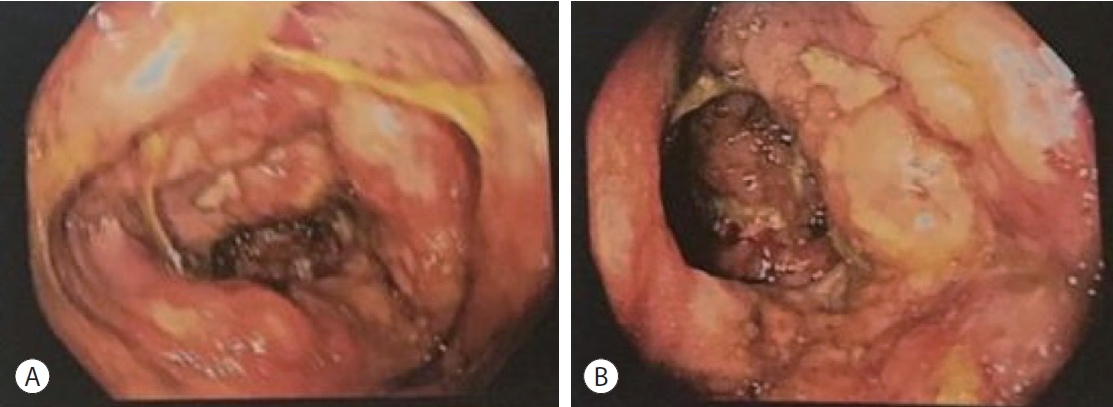
- Colonoscopy revealed a deformed and edematous ascending colon and cecum, with ulcers measuring up to 1 cm in diameter (Fig. 4). Multiple biopsies were obtained from the ulcerated lesions. Histopathological examination revealed an ulcerated surface covered with an acute inflammatory exudate containing eosinophils. A Splendore-Hoeppli phenomenon was observed around the fungal spores. Periodic acidSchiff (PAS) and Grocott methenamine silver (GMS) stains highlighted the presence of fungal spores (Fig. 5).
- Based on the histology and fungal culture studies, a final diagnosis of basidiobolomycosis was made. Thus, surgical resection was deferred, and the patient was placed on oral voriconazole 6 mg/kg 12 hourly. The patient is doing currently well at 2 years follow-up visit. A multiphase follow-up contrast-enhanced CT scan demonstrated interval resolution of the irregular ileocecal thickening and paracecal lymph nodes. Moreover, there was an interval reduction in the size of hepatic lesions (Fig. 6).
CASE REPORT
- B. ranarum is a saprophytic fungus found in soil, decomposing plant matter, animal waste, and manure. The mode of infection is through insect bites, contamination of wounds or food with soil, or use of contaminated toilet paper. A review suggested that the pathogenesis of basidiobolomycosis is due to the production of lipases and proteases by the fungus.1,2 Some possible risk factors include ranitidine, a known risk factor for bacterial gastroenteritis, which decreases acidity in the stomach, promoting the survival of the organisms. Another possible risk factor is chronic smoking.3 B. ranarum is known to cause subcutaneous mycoses; subcutaneous infections have been reported mainly in the pediatric population, characterized by firm, painless nodules in the limbs, trunk, perineum, and buttocks.1
- Only a handful of cases with liver and gastrointestinal involvement have been reported.4,5-15 The colon and rectum are the most frequently reported gastrointestinal sites, and most affected people are immunocompetent.16 Liver involvement occurs as part of a disseminated infection. Clinical features and radiological findings are often non-specific and therefore pose diagnostic challenges.
- In a review, the most common abdominal CT scan finding was a colorectal mass in 48% of cases, followed by a liver mass in 30%.4 Differential diagnoses of colorectal masses include malignancy or Crohns disease in adults and tuberculosis or amebiasis in children.5 Histology and fungal culture help distinguish between these differentials. B. ranarum appears as a faintly stained, broad, sparsely septate pleomorphic fungus on GMS or PAS stains.3 The hyphae range from 5-20 µm in diameter, with a mean of 9 µm.1 Hematoxylin and eosin-stained sections show the characteristic Splendore-Hoeppli phenomenon along with eosinophilic inflammation and zygospores. Splendore-Hoeppli phenomenon is caused by the deposition of eosinophilic and hyaline materials or inert substances around organisms. This eosinophilic material is an antigenantibody complex deposited by inflammatory cells. They are commonly observed in fungal, bacterial, and parasitic infections, although they may sometimes be present in non-infective conditions such as allergic conjunctival granulomas and hypereosinophilic syndrome.6 Zygospores may appear similar to trophozoites of amebae, as they have prominent nucleoli and abundant foamy cytoplasm. Another close differential diagnosis is mucormycosis; this is associated with abundant neutrophilic infiltration unlike the eosinophilic infiltration of basidiobolomycosis, and also often demonstrates vascular invasion and necrosis. The possibility of eosinophilic gastroenteritis can be ruled out based on the presence of fungal hyphae. On culture, fungal colonies appear pale grey along with radial folds on Sabouraud agar after 2-3 days of incubation at 25-30℃.3
- Hepatic involvement most commonly mimics a carcinoma; however, as in our case, histological examination and a fungal culture can easily help distinguish between these two. So far, 19 cases of basidiobolomycoses with hepatic involvement have been reported (Table 1).7-17 The patients’ ages ranged from 23 months to 65 years, and the most common clinical presentations were abdominal pain, vomiting, weight loss, and, in some cases, an abdominal mass. The most common colonoscopic findings were obstructive masses involving the cecum and the colon. The most common CT/MRI findings were multiple lesions in the liver, along with a thickened and sometimes edematous intestinal wall. In many cases, cecal masses with or without ascending colon or terminal ileum involvement were noted. Frequent histological findings included eosinophilic infiltration, granuloma formation, the presence of broad aseptate/pauciseptate fungal elements, and Splendore-Hoeppli phenomenon. All patients received antifungal therapy, while surgery was performed in 15 patients. Most patients recovered on follow-up (16 cases), while two died of basidiobolomycosis-associated complications.
- Basidiobolomycosis is a rare fungal infection which may also present with gastrointestinal and/or hepatic involvement. As the clinical and radiological features are often nonspecific or suggest the possibility of malignancy, as in this case, its possibility should be considered in the differential diagnosis, especially in endemic regions.
DISCUSSION
-
Conflict of Interest
The authors have no conflicts of interest to disclose.
-
Ethics Statement
The ethics committee of the authors’ institution waived the need for ethics approval. Informed consent has been obtained from the patient.
-
Funding Statement
None.
-
Data Availability
Data sharing not applicable to this article as no datasets were generated or analyzed for this case report.
-
Author Contribution
Conceptualization: RS, NB, AS, DM, VV
Data curation: RS, NB, AS, DM, VV
Formal analysis: RS, NB
Investigation: RS, NB, AS, DM, VV
Methodology: RS, NB
Project administration: NB
Resources: RS, NB, AS, DM, VV
Supervision: NB
Validation: NB
Visualization: RS, NB
Writing original draft: RS, NB
Writing review & editing: RS, NB
Approval of final manuscript: all authors
Article information
Acknowledgments



| Author | Year of publication | Number of cases | Type of lesion | Reference |
|---|---|---|---|---|
| Abduh et al. | 2022 | 1 | Cecal perforation along with liver involvement | 7 |
| Jafarpour et al. | 2021 | 1 | GI basidiobolomycosis with liver involvement | 8 |
| Omar Takrouni et al. | 2019 | 1 | Disseminated GI basidiobolomycosis along with hepatic abscess | 5 |
| Sanaei Dashti et al. | 2018 | 1 | Disseminated basidiobolomycosis with liver involvement | 9 |
| Zekavat et al. | 2017 | 1 | Colonic basidiobolomycosis with hepatic involvement | 10 |
| Geramizadeh et al. | 2015 | 1 | Isolated hepatic basidiobolomycosis | 11 |
| Ejtehadi et al. | 2014 | 1 | GI basidiobolomycosis along with liver involvement | 13 |
| Saeed et al. | 2014 | 1 | GI basidiobolomycosis along with liver dissemination | 14 |
| Hassan et al. | 2013 | 1 | Liver and GI mass | 15 |
| Al-Shanafey et al. | 2012 | 9 | Four patients had colonic and hepatic involvement, three had isolated hepatic lesions and two had disseminated infection | 16 |
| Van den Berk et al. | 2006 | 1 | Colon along with hepatic involvement |
- 1. Hussein MRA, Alqahtani AS, Alhagawi Y, Al Saglan NS, Albishi AM, Ahmed FO, et al. Primary basidiobolomycosis of the cecum in immunocompetent patients: two case reports. Gastroenterology Res 2021;14:116−124.ArticlePubMedPMC
- 2. Gugnani HC. A review of zygomycosis due to Basidiobolus ranarum. Eur J Epidemiol 1999;15:923−929.PubMed
- 3. Lyon GM, Smilack JD, Komatsu KK, Pasha TM, Leighton JA, Guarner J, et al. Gastrointestinal basidiobolomycosis in Arizona: clinical and epidemiological characteristics and review of the literature. Clin Infect Dis 2001;32:1448−1455.ArticlePubMed
- 4. Elzein F, Mursi M, Albarrag AM, Alfiaar A, Alzahrani A. Disseminated angioinvasive basidiobolomycosis with a favourable outcome. Med Mycol Case Rep 2018;22:30−34.ArticlePubMedPMC
- 5. Takrouni AO, Schammut MH, Al-Otaibi M, Al-Mulla M, Privitera A. Disseminated intestinal basidiobolomycosis with mycotic aneurysm mimicking obstructing colon cancer. BMJ Case Rep 2019;12:e225054.ArticlePubMedPMC
- 6. Gopinath D. Splendore-Hoeppli phenomenon. J Oral Maxillofac Pathol 2018;22:161−162.ArticlePubMedPMC
- 7. Abduh MS, Aldaqal SM, Almaghrabi J, Aljiffry MM, Elbadrawy HA, Alsahafi MA. A very rare basidiobolomycosis case presented with cecal perforation and concomitant hepatic involvement in an elderly male patient: a case study. Int J Environ Res Public Health 2022;19:3412. ArticlePubMedPMC
- 8. Jafarpour Z, Pouladfar G, Dehghan A, Anbardar MH, Foroutan HR. Case report: gastrointestinal basidiobolomycosis with multi-organ involvement presented with intussusception. Am J Trop Med Hyg 2021;105:1222−1226.ArticlePubMedPMC
- 9. Dashti AS, Nasimfar A, Khorami HH, Pouladfar G, Kadivar MR, Geramizadeh B, et al. Gastro-intestinal basidiobolomycosis in a 2-year-old boy: dramatic response to potassium iodide. Paediatr Int Child Health 2018;38:150−153.ArticlePubMed
- 10. Zekavat OR, Abdolkarimi B, Pouladfar G, Fathpour G, Mokhtari M, Shakibazad N. Colonic basidiobolomycosis with liver involvement masquerading as gastrointestinal lymphoma: a case report and literature review. Rev Soc Bras Med Trop 2017;50:712−714.ArticlePubMed
- 11. Geramizadeh B, Dashti AS, Kadivar MR, Kord S. Isolated hepatic basidiobolomycosis in a 2-year-old girl: the first case report. Hepat Mon 2015;15:e30117.ArticlePubMedPMC
- 12. Flicek KT, Vikram HR, De Petris GD, Johnson CD. Abdominal imaging findings in gastrointestinal basidiobolomycosis. Abdom Imaging 2015;40:246−250.ArticlePubMedPDF
- 13. Ejtehadi F, Anushiravani A, Bananzadeh A, Geramizadeh B. Gastrointestinal basidiobolomycosis accompanied by liver involvement: a case report. Iran Red Crescent Med J 2014;16:e14109.ArticlePubMedPMC
- 14. Saeed MA, Al Khuwaitir TS, Attia TH. Gastrointestinal basidiobolomycosis with hepatic dissemination: a case report. JMM Case Rep 2014;1:e003269.ArticlePubMedPMC
- 15. Hassan HA, Majid RA, Rashid NG, Nuradeen BE, Abdulkarim QH, Hawramy TA, et al. Eosinophilic granulomatous gastrointestinal and hepatic abscesses attributable to basidiobolomycosis and fasciolias: a simultaneous emergence in Iraqi Kurdistan. BMC Infect Dis 2013;13:91. ArticlePubMedPMCPDF
- 16. Al-Shanafey S, AlRobean F, Bin Hussain I. Surgical management of gastrointestinal basidiobolomycosis in pediatric patients. J Pediatr Surg 2012;47:949−951.ArticlePubMed
- 17. van den Berk GE, Noorduyn LA, van Ketel RJ, van Leeuwen J, Bemelman WA, Prins JM. A fatal pseudo-tumour: disseminated basidiobolomycosis. BMC Infect Dis 2006;6:140. ArticlePubMedPMCPDF
References
Figure & Data
References
Citations


 E-submission
E-submission THE KOREAN LIVER CANCER ASSOCIATION
THE KOREAN LIVER CANCER ASSOCIATION
 PubReader
PubReader ePub Link
ePub Link Download Citation
Download Citation

 Follow JLC on Twitter
Follow JLC on Twitter