Search
- Page Path
- HOME > Search
Review Article
- The role of lenvatinib in the era of immunotherapy of hepatocellular carcinoma
- Matthew Man Pok Lee, Landon Long Chan, Stephen Lam Chan
- J Liver Cancer. 2023;23(2):262-271. Published online August 17, 2023
- DOI: https://doi.org/10.17998/jlc.2023.07.17
- 3,531 Views
- 281 Downloads
- 5 Citations
-
 Abstract
Abstract
 PDF
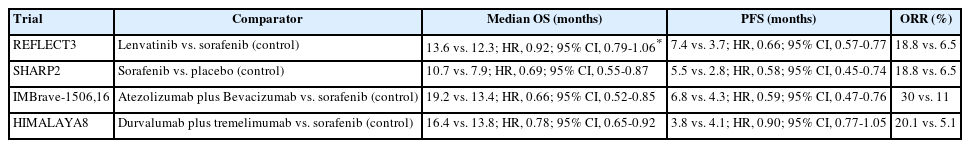
PDF - Hepatocellular carcinoma (HCC) frequently presents as advanced stage with poor prognosis and high mortality. Systemic treatment is the treatment of choice for advanced disease. In 2007, the first multi-kinase inhibitor (MKI) sorafenib was approved and shown to modestly prolong overall survival (OS). The progress of systemic therapy has been slow afterwards until 2018 when lenvatinib, another MKI, was shown to be non-inferior to sorafenib on median OS as the first-line therapy for HCC. Since then, remarkable progress has been achieved on the treatment of advanced HCC, including the development of second-line targeted treatment, including regorafenib, cabozantinib and ramucirumab from 2017 to 2019. A growing focus has been placed on immune checkpoint inhibitors (ICIs) targeting programmed cell death-1 (PD-1), its ligand PD-L1, and cytotoxic T-lymphocyte-associated protein 4. These ICIs have proven their potency in treating HCC as both initial and subsequent line of therapy. At present, both regimens of atezolizumab combined with bevacizumab, as well as the combination of tremelimumab and durvalumab, are recommended as the first-line treatments based on positive phase III clinical trials. With the advancement of ICIs, it is anticipated that the role of MKIs in the treatment of HCC will evolve. In this article, lenvatinib, one of the most commonly used MKIs in HCC, is chosen to be reviewed.
-
Citations
Citations to this article as recorded by- Reduced-Dose or Discontinuation of Bevacizumab Might Be Considered after Variceal Bleeding in Patients with Hepatocellular Carcinoma Receiving Atezolizumab/Bevacizumab: Case Reports
Kyeong-Min Yeom, Young-Gi Song, Jeong-Ju Yoo, Sang Gyune Kim, Young Seok Kim
Medicina.2024; 60(1): 157. CrossRef - The Position of Multikinase Inhibitors in the Era of Immune-Checkpoint Inhibitors for Hepatocellular Carcinoma
Beom Kyung Kim
Gut and Liver.2024; 18(1): 3. CrossRef - Fatal intratumoral hemorrhage in a patient with hepatocellular carcinoma following successful treatment with atezolizumab/bevacizumab: A case report
Kyeong-Hoon Park, Jeong-Ju Yoo, Sang Gyune Kim, Young Seok Kim
World Journal of Clinical Cases.2024; 12(22): 5177. CrossRef - Small molecule tyrosine kinase inhibitors approved for systemic therapy of advanced hepatocellular carcinoma: recent advances and future perspectives
Jianzhong Liu, Shuai Xia, Baoyi Zhang, Dina Mostafa Mohammed, Xiangliang Yang, Yanhong Zhu, Xinnong Jiang
Discover Oncology.2024;[Epub] CrossRef - Consistent efficacy of hepatic artery infusion chemotherapy irrespective of PD‑L1 positivity in unresectable hepatocellular carcinoma
Ji Kim, Young Kim, Hee-Chul Nam, Chang-Wook Kim, Jae-Sung Yoo, Ji Han, Jeong Jang, Jong Choi, Seung Yoon, Ho Jong Chun, Jung Oh, Suho Kim, Sung Lee, Pil Sung
Oncology Letters.2024;[Epub] CrossRef
- Reduced-Dose or Discontinuation of Bevacizumab Might Be Considered after Variceal Bleeding in Patients with Hepatocellular Carcinoma Receiving Atezolizumab/Bevacizumab: Case Reports

Case Report
- A case report of a patient presented with skin ulcer after treatment of lenvatinib
- Serin Cha, Dong Woo Kim, Jung Wan Choe, Tae Hyung Kim, Seung Young Kim, Jong Jin Hyun, Sung Woo Jung, Ja Seol Koo, Young Kul Jung, Hyung Joon Yim
- J Liver Cancer. 2021;21(2):194-198. Published online September 30, 2021
- DOI: https://doi.org/10.17998/jlc.2021.09.20
- 3,635 Views
- 86 Downloads
- 2 Citations
-
 Abstract
Abstract
 PDF
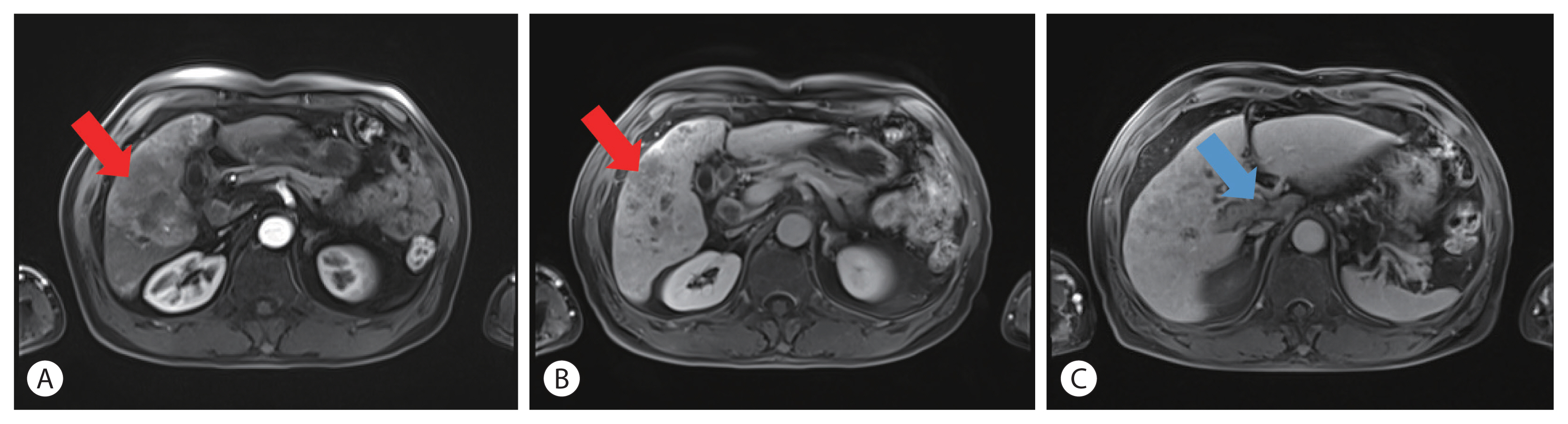
PDF - A 60-year-old man diagnosed with unresectable hepatocellular carcinoma (HCC) presented to the hospital with pain in the perineal region. He had been taking lenvatinib every day for 2 months after he was diagnosed with HCC with metastases to the lymph node, small bowel mesentery, and retroperitoneal space. Enhanced abdominal computed tomography revealed mild elevation in intensity in the perineal subcutaneous tissue with subcutaneous emphysema. The patient was diagnosed with Common Terminology Criteria for Adverse Events grade 3, skin ulceration of stage IV with full-thickness skin loss and tissue necrosis in the muscular layer. The patient was taken off the medication with prescription of antibiotics, and after 3 weeks, the skin has fully recovered. This is the first report of an HCC patient who presented with a skin ulceration of stage IV after lenvatinib treatment. We recommend stopping the medication immediately and changing to alternative treatments with appropriate supportive care.
-
Citations
Citations to this article as recorded by- Multiple lenvatinib‐associated skin ulcers: A case report and literature review
Soo Hyun Jeon, Woo Jin Lee, Chong Hyun Won, Sung Eun Chang, Mi Woo Lee, Joon Min Jung
Australasian Journal of Dermatology.2023;[Epub] CrossRef - Tyrosine kinase inhibitors induced scrotal ulcerations: Report of 2 cases
Abhipsa Samal, Nibedita Dixit, BikashR Kar, Liza Mohapatra
Indian Journal of Dermatology.2023; 68(2): 235. CrossRef
- Multiple lenvatinib‐associated skin ulcers: A case report and literature review

Original Article
- Infiltration of T Cells and Programmed Cell Death Ligand 1-expressing Macrophages as a Potential Predictor of Lenvatinib Response in Hepatocellular Carcinoma
- Pil Soo Sung, Sung Woo Cho, Jaejun Lee, Hyun Yang, Jeong Won Jang, Si Hyun Bae, Jong Young Choi, Seung Kew Yoon
- J Liver Cancer. 2020;20(2):128-134. Published online September 30, 2020
- DOI: https://doi.org/10.17998/jlc.20.2.128
- 3,545 Views
- 101 Downloads
- 6 Citations
-
 Abstract
Abstract
 PDF
PDF - Background/Aims
Lenvatinib was recently proven to be non-inferior to sorafenib in treating unresectable hepatocellular carcinoma (HCC) in a phase-3 randomized controlled trial. In this study, we investigated whether the response to lenvatinib was affected by tumor immunogenicity.
Methods
Between May 2019 and April 2020, nine patients with intermediate-to-advanced HCC, who were treated with lenvatinib after liver biopsy, were enrolled. Immunohistochemical staining and multi-color flow cytometry were performed on specimens obtained from liver biopsy.
Results
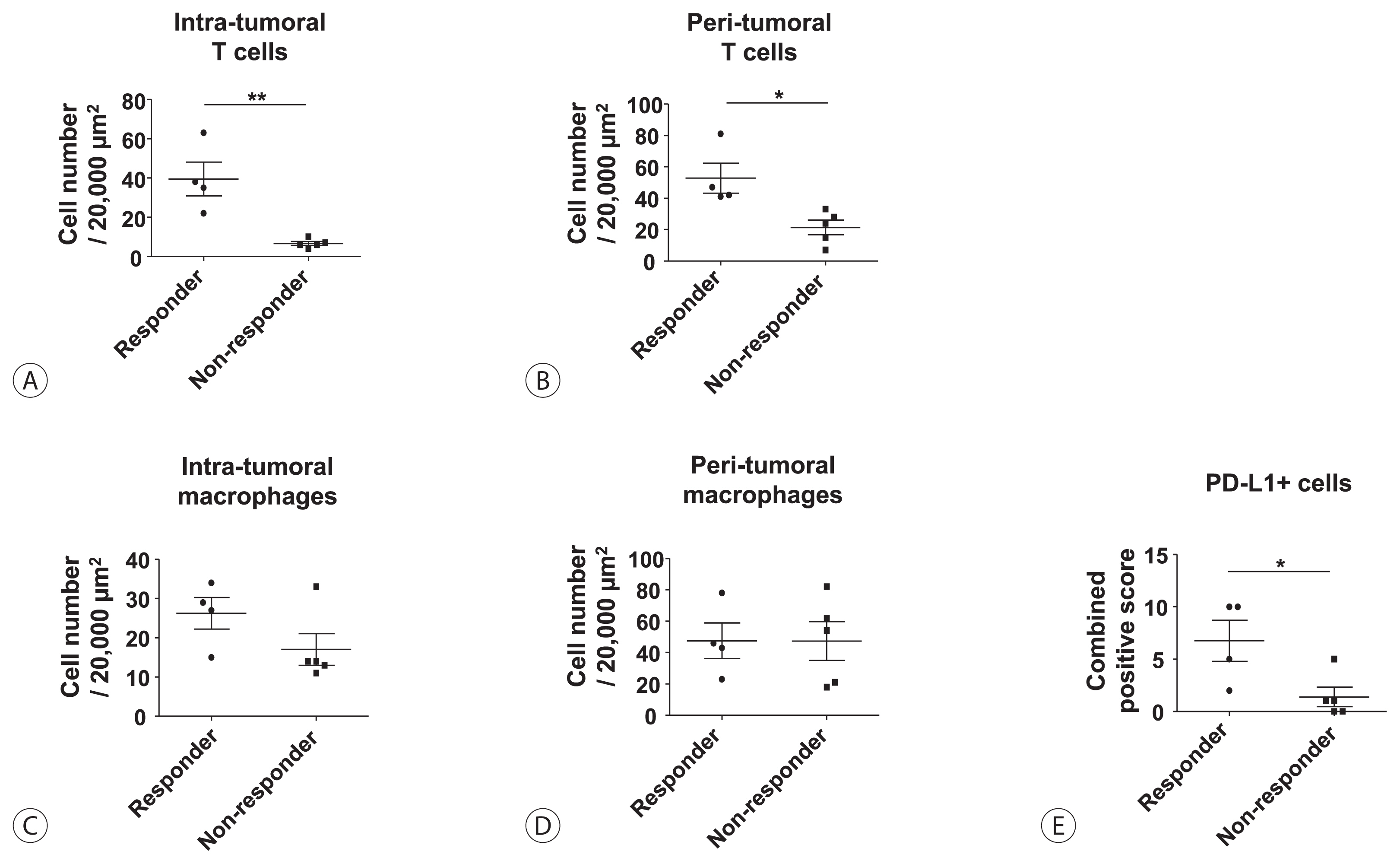
Among the nine patients enrolled, four showed objective responses (complete responses+partial responses). Immunohistochemical staining for CD3, CD68, and programmed cell death ligand 1 (PD-L1) demonstrated that patients with objective responses showed marked infiltration of T cells and PD-L1-expressing macrophages in intra-tumoral and peri-tumoral tissues compared to those without objective responses. A significant difference in the numbers of infiltrated T cells, both in the intra-tumoral (P<0.01) and peri-tumoral regions (P<0.05), were identified between responders and non-responders. Regarding the number of infiltrated macrophages, no significant difference was found between the responders and non-responders, although the number of PD-L1-expressing tumor-associated macrophages was significantly higher in responders than that in non-responders (P<0.05).
Conclusions
Tumor immunogenicity, as indicated by T cell and PD-L1-positive macrophage infiltration, affects lenvatinib response in unresectable HCC. -
Citations
Citations to this article as recorded by- Higher Number of Tumor-Infiltrating PD-L1+ Cells Is Related to Better Response to Multikinase Inhibitors in Hepatocellular Carcinoma
Ji Won Han, Ji Hoon Kim, Dong Hyun Kim, Jeong Won Jang, Si Hyun Bae, Jong Young Choi, Seung Kew Yoon, Jaegyoon Ahn, Hyun Yang, Pil Soo Sung
Diagnostics.2023; 13(8): 1453. CrossRef - Intrahepatic inflammatory IgA+PD-L1high monocytes in hepatocellular carcinoma development and immunotherapy
Pil Soo Sung, Dong Jun Park, Pu Reun Roh, Kyoung Do Mun, Sung Woo Cho, Gil Won Lee, Eun Sun Jung, Sung Hak Lee, Jeong Won Jang, Si Hyun Bae, Jong Young Choi, Jonghwan Choi, Jaegyoon Ahn, Seung Kew Yoon
Journal for ImmunoTherapy of Cancer.2022; 10(5): e003618. CrossRef - Crosstalk between tumor-associated macrophages and neighboring cells in hepatocellular carcinoma
Pil Soo Sung
Clinical and Molecular Hepatology.2022; 28(3): 333. CrossRef - Blood-based biomarkers for immune-based therapy in advanced HCC: Promising but a long way to go
Pil Soo Sung, Isaac Kise Lee, Pu Reun Roh, Min Woo Kang, Jaegyoon Ahn, Seung Kew Yoon
Frontiers in Oncology.2022;[Epub] CrossRef - Immunological Mechanisms for Hepatocellular Carcinoma Risk after Direct-Acting Antiviral Treatment of Hepatitis C Virus Infection
Pil Soo Sung, Eui-Cheol Shin
Journal of Clinical Medicine.2021; 10(2): 221. CrossRef - Preferential Expression of Programmed Death Ligand 1 Protein in Tumor-Associated Macrophages and Its Potential Role in Immunotherapy for Hepatocellular Carcinoma
Dong-Jun Park, Pil-Soo Sung, Gil-Won Lee, Sung-Woo Cho, Sung-Min Kim, Byung-Yoon Kang, Won-Hee Hur, Hyun Yang, Soon-Kyu Lee, Sung-Hak Lee, Eun-Sun Jung, Chang-Ho Seo, Joseph Ahn, Ho-Joong Choi, Young-Kyoung You, Jeong-Won Jang, Si-Hyun Bae, Jong-Young Cho
International Journal of Molecular Sciences.2021; 22(9): 4710. CrossRef
- Higher Number of Tumor-Infiltrating PD-L1+ Cells Is Related to Better Response to Multikinase Inhibitors in Hepatocellular Carcinoma


 E-submission
E-submission THE KOREAN LIVER CANCER ASSOCIATION
THE KOREAN LIVER CANCER ASSOCIATION

 First
First Prev
Prev



 Follow JLC on Twitter
Follow JLC on Twitter