Imaging Modalities for Hepatocellular Carcinoma Surveillance: Expanding Horizons beyond Ultrasound
Article information
Abstract
In Asian countries favoring loco-regional treatment such as surgical resection or ablation, very early-stage hepatocellular carcinoma (HCC) should be the main target for surveillance. Even though ultrasound (US) has been accepted as a primary imaging modality for HCC surveillance, its performance in detecting very early-stage HCCs is insufficient. Moreover, in more than 20% of patients at high risk for HCC, visualization of the liver on US may be limited owing to the advanced distortion and heterogeneity of the liver parenchyma. Recently revised HCC clinical guidelines allow the use of alternative surveillance tools including computed tomography or magnetic resonance imaging in patients with inadequate US exams. This paper summarizes the findings of recent studies using imaging modalities other than US as surveillance tools for HCC as well as strengths and limitations of these modalities.
INTRODUCTION
Hepatocellular carcinoma (HCC) is the leading cause of death in patients with liver cirrhosis, largely due to failed early detection.1 Therefore, international guidelines recommend imaging-based HCC surveillance for patients at high risk.2–4 The treatment patterns for HCC vary across regions or countries according to the demographic characteristics, HCC prevalence, socioeconomic status, and medical resource availability.5 In Asian countries including Korea, surgical resection and image-guided ablation are preferred over deceased donor liver transplantation (DDLT), which is the main curative treatment option in Western countries. In this setting favoring loco-regional treatment, the main target for HCC surveillance differs from that in settings in which liver transplantation, especially DDLT, is the primary option. While DDLT as an HCC treatment requires high specificity comparable to that attained by histopathologic diagnosis to maximize organ utilization,6 loco-regional treatment emphasizes the importance of sensitive detection of HCC at very early stage. The outcomes of surgical resection7,8 and ablation9–11 are closely related to HCC size, with 2 cm suggested as a size cut-off for HCC aggressiveness and invasiveness.7,12,13 Especially for radiofrequency ablation, the preferable size is smaller than 2 cm11 as the goal is to obtain a 360-degree, 0.5–1.0 cm ablative margin all around the target tumor.14 Thus, in Asian countries favoring loco-regional treatment, very early-stage HCC should be the main target for surveillance. The goal of this article was to review the performance and limitations of ultrasound (US), to update readers regarding recent studies assessing imaging modalities other than US as possible HCC surveillance tools, and to summarize the strengths and limitations of each modality in detecting very early-stage HCC.
ULTRASOUND: LIMITATIONS AS THE SOLE IMAGING MODALITY FOR HCC SURVEILLANCE
Grayscale US is accepted as a primary imaging modality for HCC surveillance. Current clinical guidelines endorse surveillance using US with or without measurement of levels of serum tumor markers such as alpha-fetoprotein (AFP) every six months in populations at risk for HCC.2–4 However, it should be noted that US has limited sensitivity.15 A meta-analysis including 13 studies and 1,514 patients reported a US sensitivity of 63% and 69% for US with AFP in detecting early-stage HCC (one nodule <5 cm or three nodules each <3 cm in diameter) in patients with cirrhosis.16 Another recent meta-analysis with 15 studies and 4,400 patients reported sensitivity of US as low as 47% for early-stage HCC.17 For HCC smaller than 2 cm, the sensitivity of US is further decreased; a recent prospective study reported a strikingly low US sensitivity for very early-stage HCC of 27.3%.18 Given these disappointing results, a more sensitive surveillance tool is required to detect HCC at very early stages. Moreover, visualization of the liver and focal hepatic lesions on US differs between patients. Approximately 20–30% of US examinations are classified as inadequate for HCC surveillance in patients at high risk of developing HCC, and inadequate US were significantly associated with advanced liver cirrhosis.19,20 The sensitivity of US to detect hepatic lesions is also affected by lesion size and patient body status. A previous study21 showed that US sensitivity differed according to HCC size, as 85% for lesions >4 cm, 65% for lesions 2–4 cm, and 21% for lesions <2 cm. As obesity poses challenges for US to detect HCC due to limited sonic beam penetration, the sensitivity of US was only 21% in patients with body mass index (BMI) ≥30 kg/m2 compared to 77% in those with BMI <30 kg/m2.22
To address these problems, recently revised clinical HCC guidelines have allowed the use of alternative surveillance tools such as computed tomography (CT) and magnetic resonance imaging (MRI) in patients with inadequate surveillance on US.2–4 Recent studies have explored the potentials of CT and MRI with various techniques as alternative tools for HCC surveillance.
IMAGING MODALITIES BEYOND US
1. CT
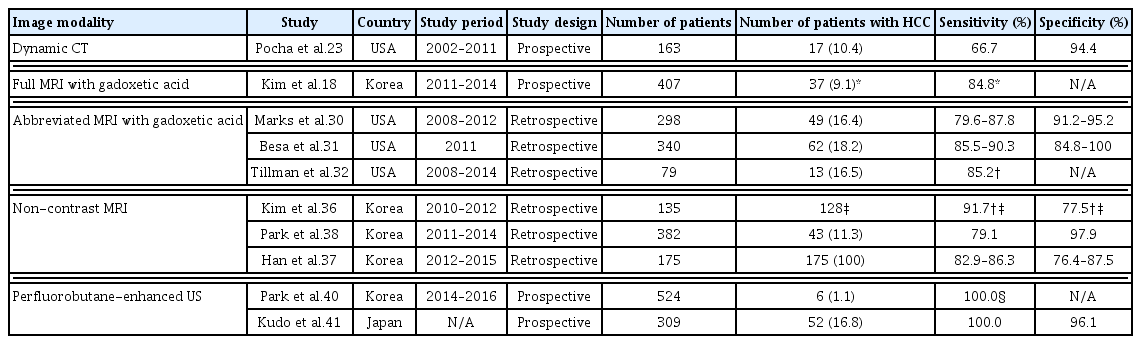
CT for HCC surveillance is yet to be established due to the paucity of data. To our knowledge, only one prospective study23 has evaluated contrast-enhanced CT as an alternative surveillance tool by comparing its performance to that of US in 163 patients with 17 HCCs. The study concluded that biannual US was marginally more sensitive (71.4%) and less costly compared to annual CT (66.7%) for the detection of HCC. However, the limitations of that study included its unclear positivity criteria for each imaging modality, that only 10 of the 17 HCCs were early-stage, and the small number of patients and HCCs. An on-going prospective trial (A Prospective Cohort Study to Compare Contrast Enhanced Liver CT & US for HCC Surveillance in High-risk Group of HCC. ClinicalTrials.gov NCT02675920) comparing contrast-enhanced CT to US for HCC surveillance in high-risk populations will hopefully provide valuable information regarding CT-based HCC surveillance. In a diagnostic setting, a recent meta-analysis24 reported an inferior sensitivity of CT to that of MRI (66% vs. 82%), while the specificity of both modalities did not differ significantly (CT vs. MRI, 92% vs. 91%). Although performances in a diagnostic setting cannot be directly applied in a surveillance setting, CT performance is expected to be lower than that of MRI for surveillance purpose. Regardless of the performance of CT for this purpose, due to the repetitive nature of surveillance examinations, the cumulated radiation hazard and potential risks related to the use of iodine contrast agents remain significant hurdles for the use of CT in a surveillance setting.
2. MRI
1) Full MRI with gadoxetic acid
Gadoxetic acid-enhanced MRI showed superb diagnostic performance in diagnosing small HCCs (2–3 cm).25–27 Gadoxetic acid-enhanced MRI with full sequences provides not only hemodynamic information as does extracellular contrast agents but also provides additional hepatobiliary phase (HBP) images which maximize focal lesion detection. According to a study comparing US and gadoxetic acid-enhanced MRI, the sensitivity of MRI (84.8%) was significantly higher than that of US (27.3%) (P <0.001) for detecting very early-stage HCC.18 Although gadoxetic acid-enhanced MRI with full sequences may be the best option from a performance point of view, the long acquisition time and high cost hamper its widespread use.
The diagnostic performance of gadoxetic acid-enhanced MRI may be affected by technical challenges and patient liver function. The optimal arterial phase images in liver MRI using gadoxetic acid are trickier to obtain compared to liver MRI using extracellular contrast agents because of the small administered volume and gadolinium content as well as the occurrence of transient severe motion.28 Hepatic parenchymal enhancement can become heterogeneous and diminished in patients with compromised liver function, which affects lesion conspicuity during the HBP.29 Thus, these could mitigate the sensitivity of MRI and also produce pseudolesions. To our knowledge, little has been reported about the effects of these factors on the performance of surveillance tests.
2) Abbreviated MRI with gadoxetic acid or extracellular contrast agent
To reduce scan time and sequences, abbreviated MRI (AMRI) protocols comprising only selected sequences such as the HBP of gadoxetic acid-enhanced MRI30–32 or dynamic contrast-enhanced sequences with extracellular contrast agents-enhanced MRI33 are currently areas of active research. AMRI with gadoxetic acid usually includes HBP, T1-weighted sequence, and diffusion-weighted imaging (DWI) for HCC detection, with T2-weighted sequences to improve specificity by excluding cysts or hemangiomas. AMRI with gadoxetic acid has a sensitivity of 80–90% and specificity of 91–98% for detecting HCC.30–32 However, the results of these studies should be interpreted with caution. As the incidence of HCC in these studies was unrealistically high (up to 18.2%), there may have been selection bias in the study populations. An ongoing prospective trial in Korea aims to determine the clinical feasibility of AMRI with gadoxetic acid compared to US (Clinical Feasibility of Abbreviated MRI for HCC Surveillance in High-risk Group, ClinicalTrials.gov NCT03731923). However, if a lesion is detected on AMRI with gadoxetic acid, the lack of hemodynamic information necessities further examination with an additional contrast-enhanced study with multiphase dynamic images to confirm whether the detected lesion is HCC. To avoid this additional imaging study, a retrospective study33 proposed a different approach using AMRI with extracellular agent containing only dynamic sequences, showing only 5% of changes in Liver Reporting & Data System (LI-RADS) categorization based on interpretation only of AMRI with extracellular agent compared to that of full sequence MRI. AMRI with extracellular contrast agent required only 7–10 minutes of scan time, suggesting its potential usefulness. However, the study was limited as it did not report the diagnostic performances for very early-stage HCC and only reported changes in LI-RADS categorization. Similar to full MRI with gadoxetic acid, the image quality of AMRI using gadoxetic acid may be lower in patients with poor liver function, affecting the diagnostic performance of this modality; furthermore, little has been reported about the effects of these factors on surveillance tests.29
3) Increased awareness of the potential risks related to gadolinium-based MRI contrast agent
Although gadolinium-based MRI contrast agents are safer than the iodinated contrast agents used in CT, recent studies have warned that, in addition to nephrogenic systemic fibrosis, gadolinium also accumulates in body tissue after repeated exposure.34,35 Thus, the use of a gadolinium contrast agent, whether in full MRI or AMRI, bears potential risks related to the retention of gadolinium in human tissue.
4) Non-contrast MRI
Non-contrast MRI is free of gadolinium-related concerns. Non-contrast MRI composed of DWI and T2-weighted imaging may also be a promising option36,37 for HCC surveillance. DWI has shown excellent performance in detecting HCC, while T2-weighted imaging allows the exclusion of common benign lesions such as hemangiomas or cysts. The combination of DWI and T2-weighted imaging may be a practical surveillance strategy for HCC. A study comparing the performance of non-enhanced MRI and US for HCC surveillance in high-risk patients38 showed significantly higher per-lesion and per-exam sensitivities for non-enhanced MRI (77.1% and 79.1%, respectively) compared to those for US (25.0% and 27.9%, respectively, P <0.001). In this study, non-contrast MRI also showed significantly higher specificity than that of US (97.9% vs. 94.5%, respectively, P <0.001). Two prospective studies in Korea are currently evaluating the usefulness of non-contrast MRI, namely, the Annual MRI Versus Biannual US for Surveillance of HCC in liver cirrhosis (MAGNUS-HCC, ClinicalTrials.gov NCT02551250) and Usefulness of Non-contrast MRI versus Non-Contrast US for Surveillance of HCC (MIRACLE-HCC, ClinicalTrials.gov NCT02514434).
PERFLUOROBUTANE-ENHANCED US
The introduction of a new US contrast agent, perfluorobutane gas-containing microbubbles (Sonazoid; GE Healthcare, Oslo, Norway), provides new opportunities for the use of contrast-enhanced US as a surveillance tool for HCC.39 It allows stable post-vascular or Kupffer phase imaging for more than 60 minutes as well as vascular phase imaging, which provides sufficient time to scan the whole liver. Two prospective studies tested the performance of perfluorobutane-enhanced US40,41 in a surveillance setting, with contradicting results. One study from Korea40 reported that the detection rate of early-stage HCC was not significantly improved by adding perfluorobutane-enhanced US to conventional B-mode US (difference, 0.4%; P =0.16), although the false referral rate was significantly reduced (difference, −3.2%; P <0.001). In contrast, a study from Japan41 suggested that perfluorobutane-enhanced US was useful for the early detection of HCC by showing significantly smaller HCC on contrast-enhanced US (13.0 mm) than that detected by conventional B-mode US (16.7 mm, P =0.011). In addition, the inherent limitations of conventional B-mode US on poor sonic window for advanced cirrhotic liver cannot be completely mitigated on contrast-enhanced US. Perfluorobutane-enhanced US may be an option for HCC surveillance among advanced imaging modalities, although additional studies are warranted to support its usefulness.
COST-EFFECTIVENESS
In a surveillance setting, the availability and cost-effectiveness of these advanced imaging modalities also require consideration.42 The key factors determining their cost-effectiveness for cancer screening include not only the cost of diagnostic tests but also the diagnostic performances of imaging studies, the incidence of HCC, and the amount of survival gained by treatment.43–45 Two studies demonstrated that risk-stratified HCC surveillance strategies with MRI targeting high- and intermediate-risk patients with cirrhosis were cost-effective and outperformed the currently recommended non-stratified biannual US in all patients with cirrhosis.46,47 Goossens et al.46 reported the lowest incremental cost-effective ratio of $2,100 per quality-adjusted life-year for the use of AMRI for high- and intermediate-risk patients without screening of low-risk patients. Another study using Markov modeling suggested that full gadoxetic acid-enhanced MRI might be a cost-effective surveillance option in high-risk patients with an annual HCC incidence rate of 1.81% or higher, with an incremental cost-effective ratio of less than $50,000 per quality-adjusted life-year.47
CONCLUSIONS
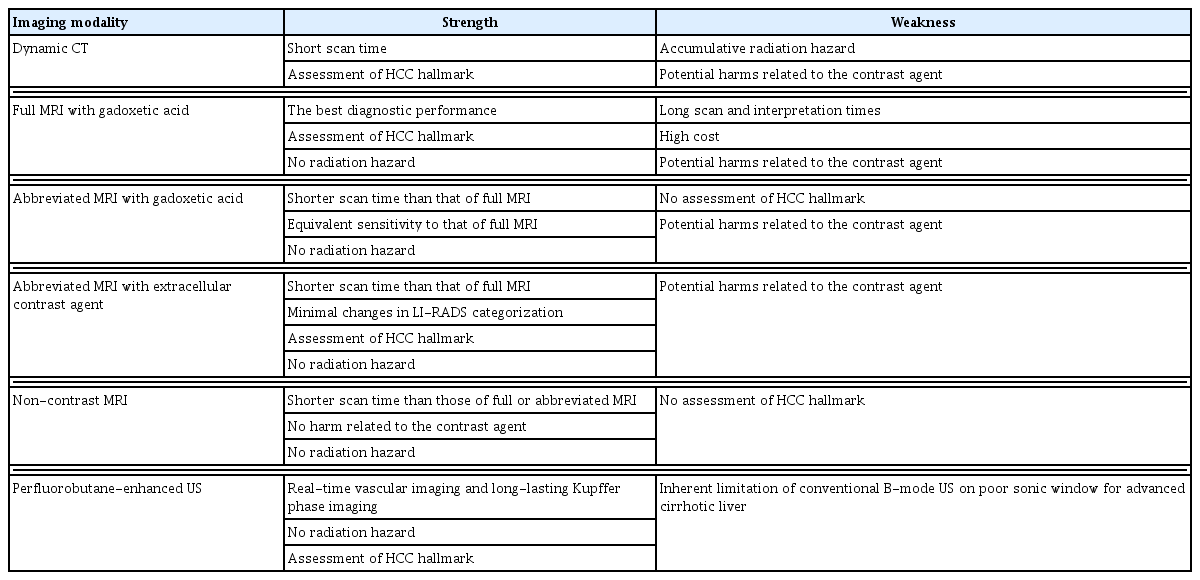
Evidence of the clinical feasibility of advanced imaging modalities other than US is rapidly accumulating owing to efforts to overcome the limitations of US for HCC surveillance, especially for very early-stage HCC (Table 1). The various options in advanced imaging modalities for HCC surveillance have their strengths and weaknesses (Table 2) which should be considered when determining the most appropriate niche for these new approaches. Instead of a one-size-fits-all approach with biannual US regardless of the HCC risks of individual patients, the results of recent studies support a risk-stratified approach with advanced imaging modalities. The potential harms related to these alternative approaches and the availability of medical resources should also be considered.48
Characteristics of studies assessing the performance of advanced imaging modalities for HCC surveillance
Notes
Conflicts of Interest
The authors declare no conflicts of interest pertaining to this work.