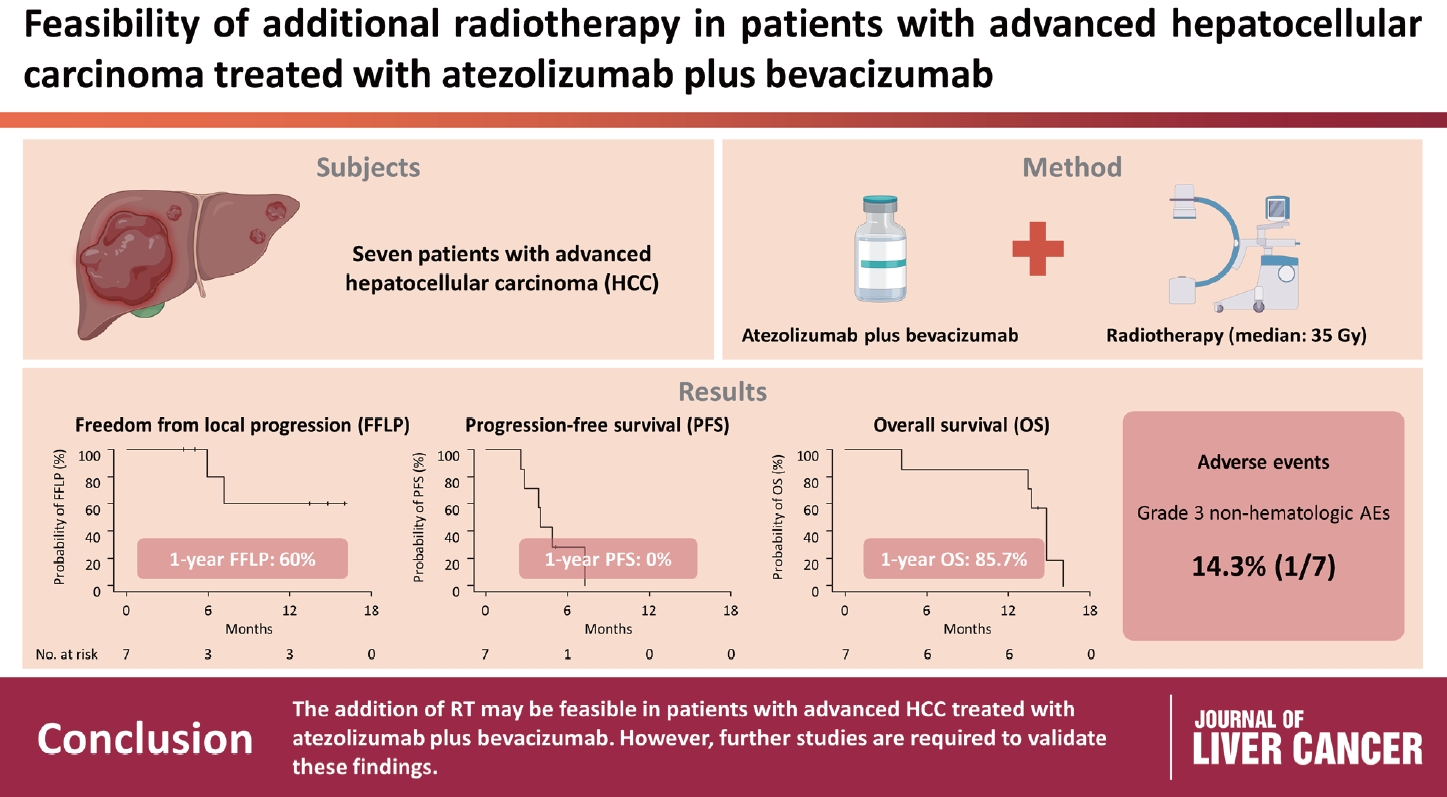
Feasibility of additional radiotherapy in patients with advanced hepatocellular carcinoma treated with atezolizumab plus bevacizumab
Article information
Abstract
Background/Aim
Radiotherapy (RT) is an effective local treatment for hepatocellular carcinoma (HCC). However, whether additional RT is safe and effective in patients with advanced HCC receiving atezolizumab plus bevacizumab remains unclear. This retrospective cohort study aimed to evaluate the feasibility of additional RT in these patients.
Methods
Between March and October 2021, we retrospectively analyzed seven patients with advanced HCC who received RT during treatment with atezolizumab plus bevacizumab. The median prescribed RT dose was 35 Gy (range, 33–66). Freedom from local progression (FFLP), progression-free survival (PFS), and overall survival (OS) after RT were analyzed.
Results
The median follow-up duration after RT was 14.2 months (range, 10.0–18.6). Of the seven patients, disease progression was noted in six (85.7%), the sites of disease progression were local in two (28.6%), intrahepatic in four (57.1%), and extrahepatic in four (57.1%). The median time of FFLP was not reached, and PFS and OS times were 4.0 (95% confidence interval [CI], 3.6–4.5) and 14.8% (95% CI, 12.5–17.2) months, respectively. The 1-year FFLP, PFS, and OS rates were 60% (95% CI, 43.8–76.2), 0%, and 85.7% (95% CI, 75.9–95.5), respectively. Grade 3 or higher hematologic adverse events (AEs) were not observed, but grade 3 nonhematologic AEs unrelated to RT were observed in one patient.
Conclusions
The addition of RT may be feasible in patients with advanced HCC treated with atezolizumab plus bevacizumab. However, further studies are required to validate these findings.
INTRODUCTION
Hepatocellular carcinoma (HCC) is the most common primary liver cancer and one of the leading causes of cancer-related deaths, primarily due to the presence of underlying chronic liver disease, late diagnosis, and frequent recurrence or progression after treatment.1,2 Treatment modalities for HCC include various local treatments, such as surgical resection, liver transplantation, radiofrequency ablation, percutaneous ethanol injection, transarterial chemoembolization, transarterial radioembolization, and radiotherapy (RT), as well as systemic treatments depending on the tumor stage, underlying liver function, and performance status.3-6 Although early stage HCC may be curable via surgical resection, liver transplantation, or ablation, most HCCs present as an advanced-stage and unresectable disease. Systemic treatments, such as those involving multikinase inhibitors (e.g., sorafenib and lenvatinib), have been used in patients with advanced and unresectable HCC despite their modest activity (i.e., modest improvement in survival [2–3 months] and low response rate [<5%]).7-9 The recent IMbrave150 trial showed significantly better survival outcomes with atezolizumab plus bevacizumab than with sorafenib.10,11 Nevertheless, prognosis remains poor in patients with advanced and unresectable HCC, even with systemic treatment, making it difficult to develop a treatment strategy without combining local and systemic treatments. Therefore, there is an unmet need for multidisciplinary treatments, that is, combinations of systemic and local treatment modalities, to improve treatment outcomes, and many studies have examined their efficacy and underlying mechanisms.12-16
RT has emerged as a promising treatment modality for localized disease that is unsuitable for curative treatment or advanced disease with macroscopic vascular invasion and/or extrahepatic disease.17-31 Recent data have shown that radiation can cause immune-induced cell death and reprogram the tumor microenvironment against the immune avoidance mechanisms of cancer. Thus, the addition of RT to combined treatment with immunotherapeutic and anti-angiogenic agents (i.e., atezolizumab and bevacizumab) could enhance treatment outcomes by creating a synergistic effect.12-16,32-34 However, the safety and efficacy of additional RT in patients with advanced HCC undergoing treatment with atezolizumab plus bevacizumab remain unclear. Thus, we retrospectively evaluated patients with advanced HCC who received additional RT during treatment with atezolizumab plus bevacizumab and assessed the feasibility of additional RT in these patients.
METHODS
1. Patients
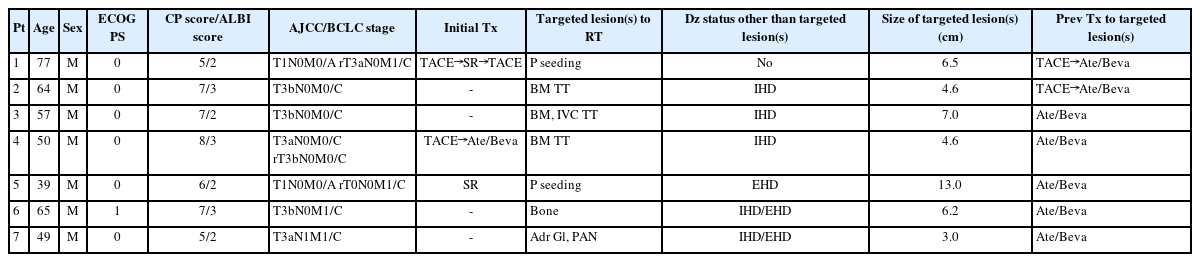
The data of patients who received additional RT for advanced HCC during treatment with atezolizumab plus bevacizumab between March and October 2021 were retrospectively reviewed. Clinical and tumor stages were classified using the Barcelona Clinic Liver Cancer3 and American Joint Committee on Cancer35 staging classification, respectively. Data from the medical records of each patient, including age, sex, performance status, tumor size, clinical and tumor stage, baseline laboratory tests (alpha-fetoprotein, albumin, and bilirubin levels, etc.), treatment before RT, details of RT (prescribed radiation dose, irradiated site[s]), treatment after RT, and times and sites of disease progression, were collected. The collected data for each patient were assigned case numbers and anonymized. Data analyses were performed according to the relevant regulations, including good clinical practice and the Declaration of Helsinki guidelines. This study was approved by the Institutional Review Board of the National Cancer Center (NCC20230014), and the requirement for written informed consent was waived owing to the retrospective study design. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines were followed (Supplementary Table 1).
2. Treatment
Standard doses of atezolizumab (1,200 mg per dose) and bevacizumab (15 mg/kg per dose) were administered to each patient every 3 weeks unless there was any specific reason for dose adjustment. The timing of RT was heterogeneous; however, patients were typically recommended the addition of RT during or at the time of treatment initiation with atezolizumab plus bevacizumab based on a multidisciplinary evaluation. RT with passive-scatter proton beam therapy or intensity-modulated RT was performed at the discretion of the physician and patient. The RT techniques, planning, and treatment procedures have been described in detail in our previous reports.21-26,28 Contrast-enhanced computed tomography (CT) was performed with each patient in the supine position, with immobilization and motion management considerations, including shallow respiration with abdominal compression or four-dimensional simulation. The gross tumor volume was defined as the tumor visualized on contrastenhanced CT images fused with dynamic CT and/or magnetic resonance images. The clinical target volume was defined as the sum of the gross tumor volume and internal target motion. Individualized planning target volume (PTV) margins of 0–7 mm from the clinical target volume were applied to compensate for respiratory motion and setup uncertainties and to ensure safe avoidance of organs at risk. Intensity-modulated RT and RT with passive-scatter proton beam therapy (Eclipse, version 13.7; Varian Medical Systems, Palo Alto, CA, USA) were performed using volumetric full or partial arcs of 6 MV X-ray and 230 MeV double-scattered proton beams (Proteus 235; Ion Beam Applications, Louvain-laNeuve, Belgium), respectively. Doses ranging from 33 to 66 Gy in 10 fractions were primarily prescribed depending on the dose-volume constraints of the organs at risk.21-26,28 During each treatment, all patients were instructed to fast for at least four hours before treatment, and the actual radiation was delivered after daily verification of the setup accuracy using either digital orthogonal X-rays or kilovoltage cone-beam CT.
3. Assessments and statistical analysis
Clinical, laboratory (including alpha-fetoprotein levels), and radiological examinations (including dynamic liver CT and magnetic resonance imaging) were performed every 2–3 months for the first 2 years and every 6 months thereafter. The albumin-bilirubin (ALBI) score was calculated using the formula: ALBI score = (log10 bilirubin [µmol/L] × 0.66) + (albumin [g/L] × -0.085), and the ALBI grades were classified based on the specific cutoff values: grade 1, ≤-2.60; grade 2, >-2.60 and ≥-1.39; and grade 3, >-1.39.36 Tumor response and disease progression were assessed based on the Response Evaluation Criteria in Solid Tumors (version 1.1)37 and RT-related adverse events (AEs) were assessed according to the Common Terminology Criteria for AEs (version 4.03). Disease progression was classified according to the site of growth or new tumors as follows: local progression within the PTV, intrahepatic progression within the liver outside the PTV, and extrahepatic progression outside the liver, such as in the regional or non-regional lymph nodes and distant organs. The time of freedom from local progression (FFLP), progression-free survival (PFS), and overall survival (OS) were determined from the commencement date of RT to the date of local progression, disease progression or death, and death from any cause or the last follow-up, respectively. The probability of survival was estimated using the Kaplan–Meier method, and all statistical analyses were performed using the STATA software (version 14.0; StataCorp, College Station, TX, USA).
RESULTS
Between March and October 2021, seven patients received additional RT during combined treatment with atezolizumab and bevacizumab at our institute. Of the seven patients, five received RT after one cycle of atezolizumab + bevacizumab, while two received RT after three and seven cycles of atezolizumab + bevacizumab, respectively. A median of seven cycles (range, 3–9) of atezolizumab plus bevacizumab treatment were continued after RT. The patient and pretreatment characteristics are summarized in Table 1. The target sites of RT were tumor thrombosis of the major vessels (n=3) and metastatic lesions (peritoneal seeding [n=2], bone [n=1], and adrenal glands and para-aortic lymph nodes [n=1]). The median prescribed dose of RT was 35 Gy (range, 33–66) in 10 and 5 fractions/week. The targeted lesion(s) and overall tumor response in the patients were as follows: complete response in one (14.3%) and one (14.3%), partial response in four (57.1%) and one (14.3%), stable disease in two (28.6%) and four (57.4%), and progressive disease in 0 (0%) and one (14.3%), respectively (Fig. 1). The median duration of follow-up for all patients after RT was 14.2 months (range, 10.0–18.6).

(A-G) Pretreatment computed tomography (CT) scans showing the target lesion(s) of radiotherapy (RT) (arrows) in each patient (No. 1–7, respectively). (H-N) RT (33–66 Gy in 10 fractions) in each patient (No. 1–7, respectively). (O-U) CT scans at 2–3 months after RT showing response of the targeted lesion(s) (arrow) in each patient (No. 1–7, respectively).
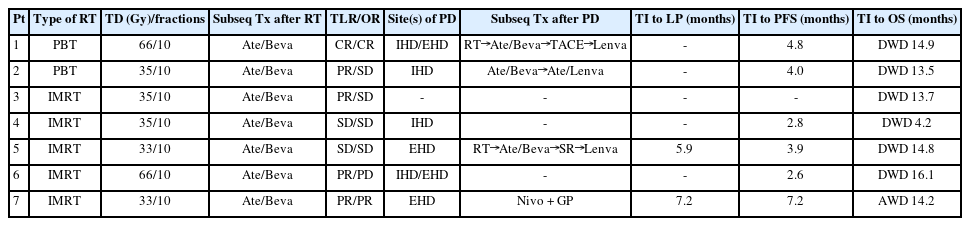
At the time of the analysis, one patient was alive, while five had died due to disease progression and one had died of an unconfirmed cause due to follow-up loss from 5 months after RT. Disease progression occurred in six of the seven (85.7%) patients, with the initial sites being local in one (14.3%), intrahepatic in three (42.9%), and extrahepatic in four (57.1%) (Table 2). The sites of disease progression were local in two (28.6%), intrahepatic in four (57.1%), and extrahepatic in four (57.1%) patients (Table 2). Two patients experienced local progression at 5.9 and 7.2 months after RT, respectively, while disease progression was observed in six patients at a median of 4.0 months (range, 2.7–4.5) after RT (Table 2). After confirmation of disease progression, five patients received one or a combination of systemic and local treatments (i.e., transarterial chemoembolization, RT, atezolizumab, lenvatinib, nivolumab, etc.), and two patients received the best supportive care, considering their performance status and willingness (Table 2). The median time of FFLP was not reached, and the PFS and OS times were 4.0 (95% confidence interval [CI], 3.6–4.5), and 14.8 months (95% CI, 12.5–17.2), respectively. The 1-year FFLP, PFS, and OS rates were 60% (95% CI, 43.8–76.2), 0%, and 85.7% (95% CI, 75.9–95.5), respectively (Fig. 2).
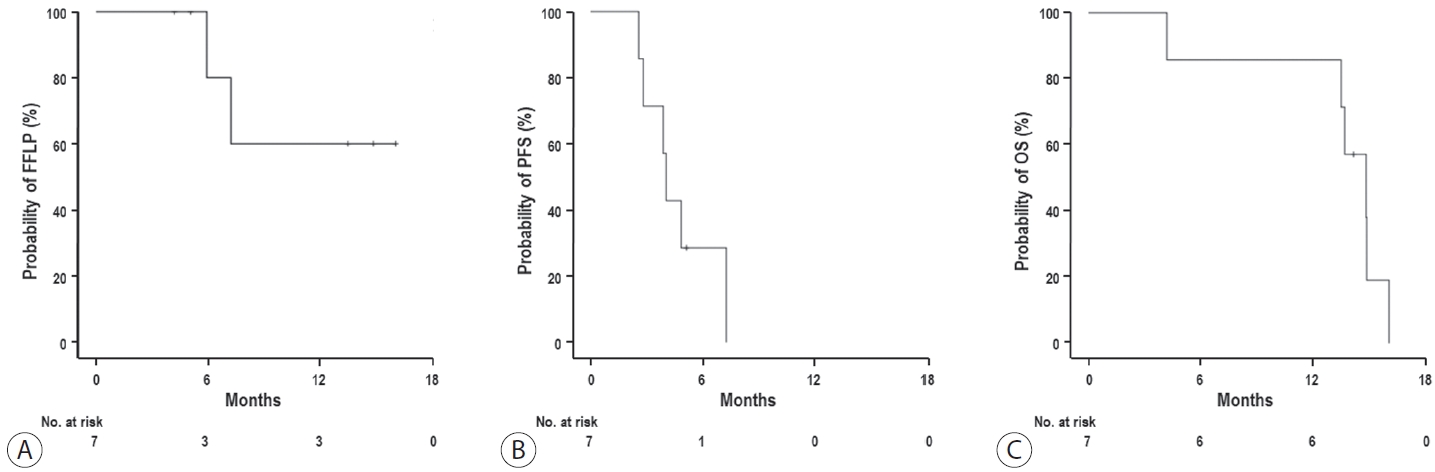
(A) Freedom from local progression (FFLP), (B) progression-free survival (PFS), and (C) overall survival (OS) curves.
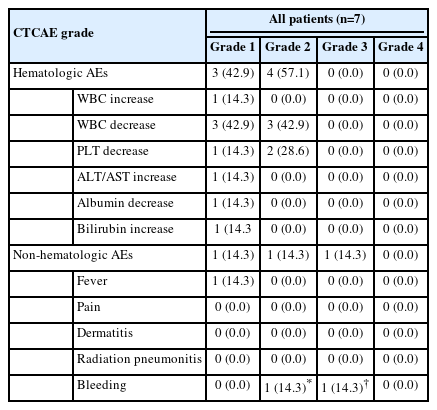
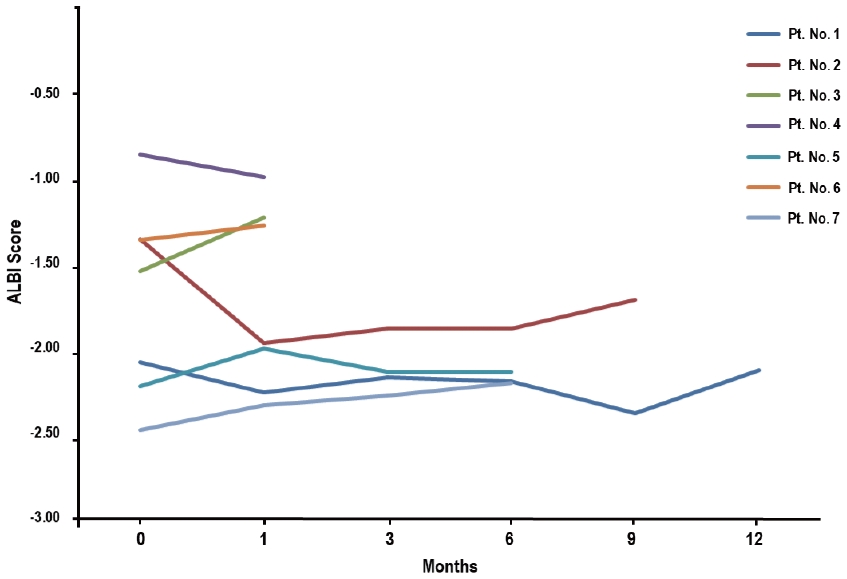
AEs during and after RT are summarized in Table 3. Among the seven patients who received additional RT during treatment with atezolizumab plus bevacizumab, all experienced hematologic AEs, with three (42.9%) experiencing grade 1 and four (47.1%) experiencing grade 2, but none experienced ≥ grade 3 hematologic AEs (Table 3). Elevated alanine aminotransferase, hypoalbuminemia, and hyperbilirubinemia levels without evidence of tumor progression were observed in one patient (14.3%), all of which were of grade 1. Of the seven patients, four (57.1%) showed no change in their Child-Pugh scores, two (28.6%) showed a 1-point decrease, and one (14.3%) showed a 1-point increase. The changes in the ALBI scores of each patient are shown in Fig. 3. Of the seven patients, five (71.4%) showed no change in the ALBI grade, one (14.3%) showed a 1-grade decrease, and one (14.3%) showed a 1-grade increase. The median change in the ALBI score was -0.04 (range, -0.61 to 0.310). Non-hematologic AEs, i.e., grade 1, fever; grade 2, epistaxis; and grade 3, gastric varix bleeding that was unrelated to the direct impact of RT as the site of bleeding was outside the RT field, were observed in one patient (14.3%), but non-hematologic AEs of grade ≥4 were not observed (Table 3).
DISCUSSION
Combination of atezolizumab and bevacizumab has emerged as the first-line treatment for patients with unresectable and extrahepatic HCC, showing superior PFS and OS rates compared to those in targeted systemic therapies, such as sorafenib.10,11 The use of local treatments, including RT, in patients with HCC with macroscopic vascular invasion and extrahepatic disease can improve tumor control and the quality of life of patients by decreasing tumor-related symptoms and morbidities.21,24,27,29,30 The combination of RT and systemic treatments, such as atezolizumab plus bevacizumab, is theoretically expected to have synergistic effects,14-16 however, it is unclear whether the addition of RT to atezolizumab plus bevacizumab treatment in patients with advanced HCC is a potentially effective treatment option because of the lack of relevant studies.12,13 Although the population in the present study was small, it showed a promising objective response rate of 71.4% and 1-year FFLP rate of 60% in targeted lesion(s) treated with RT. Furthermore, the overall objective response rate was 28.6% and disease control rate was 85.7%, which were slightly higher and comparable to those of sorafenib and atezolizumab plus bevacizumab treatments (11.9 and 27.3%, and 55.3 and 73.6%, respectively) in the IMbrave150 trial.10,11 In this trial, only patients with a Child-Pugh score of ≤6 (5 [72%] and 6 [28%]) were enrolled and 77% of the patients had advanced stage disease, such as macrovascular invasion and/or extrahepatic disease. Sorafenib and atezolizumab plus bevacizumab treatments showed median OS times of 13.4 and 19.2 months and 1-year OS rates of 54.6% and 67.2%, respectively. In the present study, 57.1% patients had a Child-Pugh score of ≥7 and 100% of patients had advanced stage disease. Additional RT in patients treated with atezolizumab plus bevacizumab showed a median OS time of 14.8 months and 1-year OS rate of 85.7%. Considering that a high proportion of patients in the present study had poor liver function and/or advanced-stage disease, the survival outcomes of additional RT in patients with advanced HCC treated with atezolizumab plus bevacizumab are promising.
When considering additional RT in patients with advanced HCC treated with atezolizumab plus bevacizumab, it is important to consider its safety and efficacy. In the present study, all-cause AEs of any grade occurred in all patients; however, ≥grade 3 AEs occurred in only one patient (14.3%). In the IMbrave150 trial,11,13 atezolizumab plus bevacizumab treatment resulted in an any-grade AEs rate of 98% and ≥grade 3 AEs rate of 63%. In addition, there are concerns regarding gastrointestinal AEs, such as bleeding, when combining RT with anti-angiogenic agents, such as bevacizumab; thus, discontinuation of anti-angiogenic agents is sometimes considered during RT.13 In the present study, although bevacizumab was not discontinued during RT, grade 3 AEs occurred in one patient (14.3%) that were unrelated to RT because the bleeding site was outside the RT field. These results suggest that additional RT may be safe in patients with HCC treated with atezolizumab plus bevacizumab. However, the present study was a retrospective analysis that included a small study population (n=7). Retrospective studies are generally likely to underestimate the incidence of AEs because of recall bias and incomplete medical records. In addition, the efficacy and toxicity of the sequence of RT and atezolizumab plus bevacizumab remain unclear; however, in this study, RT was performed immediately after one cycle of atezolizumab plus bevacizumab treatment in most patients (71.4%) on the basis of a multidisciplinary evaluation considering the potential synergies between RT and atezolizumab plus bevacizumab. Further large-scale prospective studies are needed to confirm the safety and efficacy of additional RT in patients with advanced HCC receiving atezolizumab plus bevacizumab treatment.
In conclusion, RT may be a feasible and potentially effective treatment option in patients with advanced HCC treated with atezolizumab plus bevacizumab, and the addition of RT can be well-tolerated. Further studies are required to validate these findings and assess the safety and efficacy of this treatment approach.
Notes
Conflict of Interest
The authors have no conflicts of interest to disclose.
Ethics Statement
This study was approved by the Institutional Review Board of the National Cancer Center (NCC) (IRB No. NCC20230014), and the requirement for written informed consent was waived because of the retrospective study design.
Funding Statement
This research was funded by a National Cancer Center Grant (NCC2110351) and supported by the Korean Liver Cancer Association (2022). The funding source had no role in the study design, collection, analysis, or interpretation of the data.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request. The data are not publicly available due to privacy or ethical regulations.
Author Contribution
Conceptualization: THK
Data curation: THK, BHK, YC, YK, JP
Formal analysis: THK
Funding acquisition: THK
Methodology: THK, BHK, YC, YK, JP
Project administration: THK
Writing original draft: THK
Writing review & editing: THK, BHK, YC, YK, JP
Approval of final manuscript: all authors
Supplementary Material
Supplementary data can be found with this article online https://doi.org/10.17998/jlc.2023.04.14.