Hepatocellular Carcinoma in Korea: an Analysis of the 2015 Korean Nationwide Cancer Registry
Article information
Abstract
Background/Aims
Hepatocellular carcinoma (HCC) is the sixth most common cancer and the second leading cause of cancer-related death in Korea. This study evaluated the characteristics of Korean patients newly diagnosed with HCC in 2015.
Methods
Data from the Korean Primary Liver Cancer Registry (KPLCR), a representative sample of patients newly diagnosed with HCC in Korea, were analyzed. A total of 1,558 patients with HCC registered in the KPLCR in 2015 were investigated.
Results
The median age was 61.0 years (interquartile range, 54.0-70.0 years), and men accounted for 79.7% of the subjects. Hepatitis B virus infection was the most common underlying liver disease (58.1%). According to the Barcelona Clinic Liver Cancer (BCLC) staging system, stage 0, A, B, C, and D HCCs accounted for 14.2%, 31.5%, 7.6%, 39.0%, and 7.8% of patients, respectively. Transarterial therapy (32.1%) was the most commonly performed initial treatment, followed by surgical resection (23.2%), best supportive care (20.2%), and local ablation therapy (10.7%). Overall, 34.5% of patients were treated in accordance with the BCLC guidelines: 59.2% in stage 0/A, 48.4% in stage B, 18.1% in stage C, and 71.6% in stage D. The 1-, 3-, and 5-year OS rates were 67.1%, 50.9%, and 27.0%, respectively.
Conclusions
In 2015, approximately 45% of Korean HCC cases were diagnosed at a very early or early stage, and 35% of patients underwent potentially curative initial treatment. BCLC guidance was followed in 34.5% of patients; in patients with stage B or C disease, there was relatively low adherence.
INTRODUCTION
Hepatocellular carcinoma (HCC) is the sixth most common malignancy and the second leading cause of cancer-related deaths in Korea.1-3 Cancer-related death from HCC was reported to be the highest among those aged 40-59 years, i.e., those of working age who are economically active.4 As such, the annual economic burden of HCC is the highest among all cancers in Korea.5 Therefore, it is important to accumulate and provide up-to-date accurate statistics on HCC.
The Korean Primary Liver Cancer Registry (KPLCR) is a random and representative sample of patients who are newly diagnosed with primary liver cancer in Korea. More than 95% of all cancer cases in Korea are registered in the Korean Central Cancer Registry (KCCR). Among the patients with newly diagnosed primary liver cancer in the KCCR, approximately 15% were randomly selected after stratification by region and hospital where the diagnosis was made and registered in the KPLCR. Therefore, the KPLCR can be considered to contain a representative group of patients with primary liver cancer in Korea. We previously published reports on HCC-related statistics in Korea using data from the KPLCR between 2008 and 20116 and between 2012 and 2014.7 In this study, we aimed to investigate the HCC statistics, including baseline characteristics, treatment modalities, and survival, in Korea using data from the KPLCR in 2015.
METHODS
1. Patients
Patients registered in the KPLCR between January 1, 2015, and December 31, 2015, were selected. The details of the KPLCR have been presented in our previous reports.6,7 HCC was diagnosed based on histological evidence or using dynamic computed tomography (CT) and/or magnetic resonance imaging (MRI) findings (nodule >1 cm with arterial hypervascularity and portal-/delayed-phase washout).
In 2015, a total of 1,558 patients were registered in the KPLCR. Among these patients with HCC, we excluded 1) patients who received their initial treatment more than 120 days from the date of diagnosis (n=38) and 2) patients lacking information on treatment modality (n=2). Finally, 1,518 patients with HCC were included in the treatment group. The treatment group consisted of patients who received active treatment (n=1,211) and those who received best supportive care (n=307) (Supplementary Fig. 1). Thirty-eight patients who received their initial treatment more than 120 days after the first HCC diagnosis were initially excluded from the treatment group and subsequent survival analyses because they were likely to receive treatment at a more advanced tumor stage than their initial tumor stage. The Institutional Review Board (IRB) of Seoul National University Hospital decided to waive the need for IRB approval and written informed consent (IRB No. 2204-110-1317) because the KPLCR data were collected anonymously as part of the KCCR in accordance with the Cancer Control Act.
2. Data collection and definitions
Patient information was obtained from medical records from each hospital where the diagnosis of HCC was made. Well-trained KCCR-registry data recorders from each hospital investigated the medical records. Data were extracted using a standardized case record form and validated by statisticians affiliated with the KCCR and KPLCR. Data were collected for variables including baseline characteristics such as demographic, laboratory, and tumor variables; and treatment-related factors such as treatment modalities and overall survival (OS) of the patients. All tumor characteristics were assessed using diagnostic imaging, such as dynamic CT or MRI. The modified Union for International Cancer Control (mUICC) staging system and Barcelona Clinic Liver Cancer (BCLC) staging system were adopted as staging systems. OS was measured from the date of HCC diagnosis until death from any cause. Death certificate data were obtained from the national statistical data collected by the Korean Ministry of Government Administration and Home Affairs. Individual patients’ data were identified using 13-digit unique resident registration numbers issued to all Koreans. The data cutoff date was December 31, 2020.
3. Statistical analysis
Data are presented as number (percentage [%]) or median (interquartile range [IQR]), as appropriate. The significance of differences between continuous and categorical variables was examined using Student’s t-test (or Mann-Whitney U test) and the chi-squared test (or Fisher’s exact test), respectively. The OS of patients was evaluated using the KaplanMeier method, and survival differences were analyzed using the log-rank test. All statistical analyses were conducted using SPSS ver. 21.0 (IBM Corp., Armonk, NY, USA) and R version 3.5.2 (http://www.rproject.org). Two-sided P-values <0.05 were considered to indicate statistical significance.
RESULTS
1. Baseline characteristics
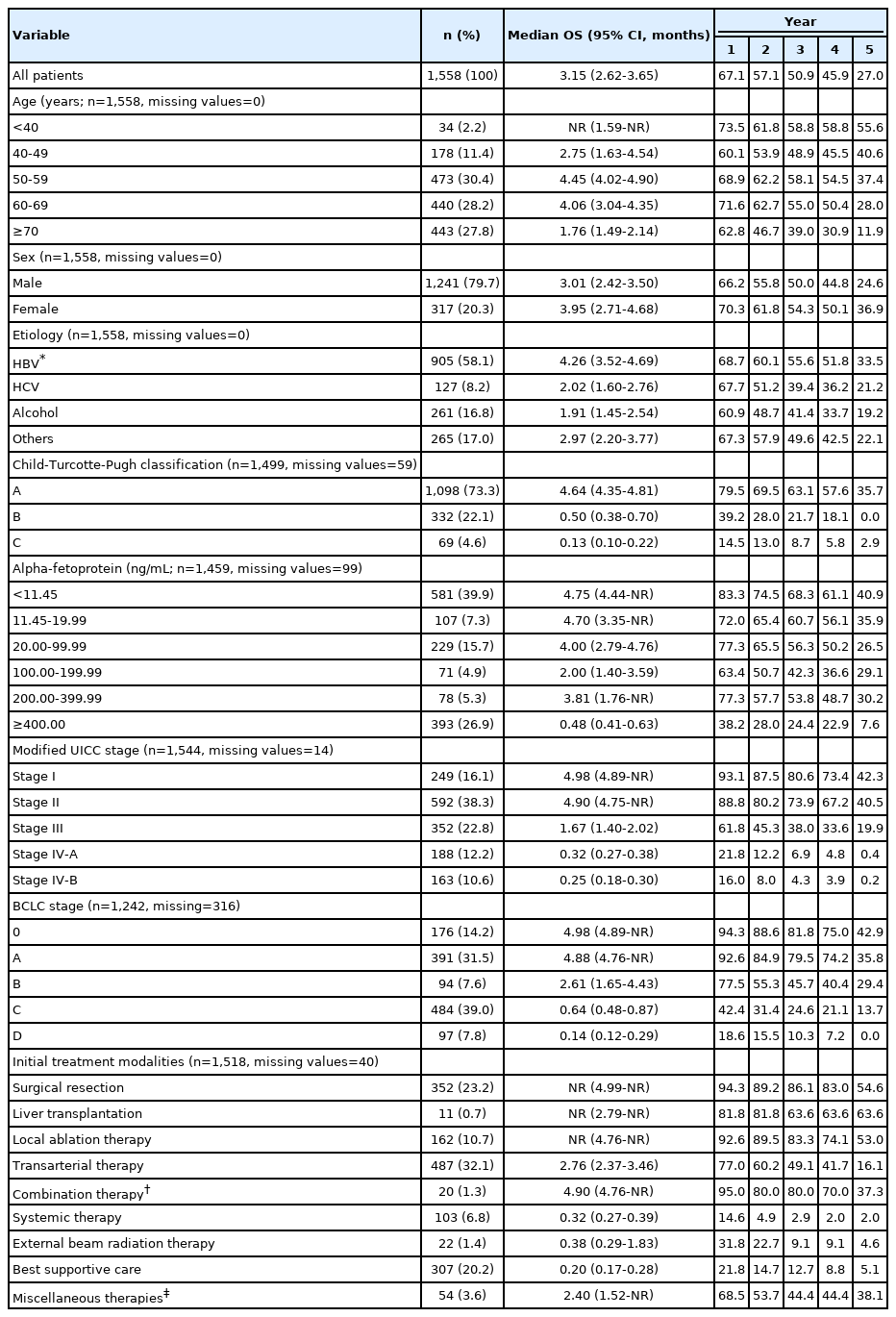
The baseline characteristics of the study population are summarized in Table 1. The median age was 61 years (IQR, 54-70 years), and the study population was predominantly male (79.7%). Hepatitis B virus (HBV) infection was the most common etiology (58.1%), followed by alcohol abuse (16.8%), hepatitis C virus (8.2%), and others (17.0%). When liver function was classified according to the Child–Turcotte–Pugh (CTP) score, 73.2%, 22.2%, and 4.6% of patients were at CTP classes A, B, and C, respectively. The median AFP level was 19.9 ng/mL (IQR, 4.7-370.3 ng/mL). According to the mUICC staging system, stages I, II, III, IV-A, and IV-B accounted for 16.1%, 38.3%, 22.8%, 12.2%, and 10.6% of patients, respectively. According to the BCLC staging system, stage C (39.0%) was the most common, followed by stage A (31.5%), stage 0 (14.2%), stage D (7.8%), and stage B (7.6%). A single tumor was observed in 61.4% of the patients and the median maximal diameter of tumors was 3.4 cm (IQR, 2.0-7.0 cm). On baseline images, portal vein and hepatic vein invasion was seen in 23.4% and 5.1% of patients, respectively. Bile duct invasion was noted in 3.1% of patients. Lymph node metastasis and distant metastasis were seen in 6.2% and 10.6% of patients, respectively.
2. Initial treatment modality
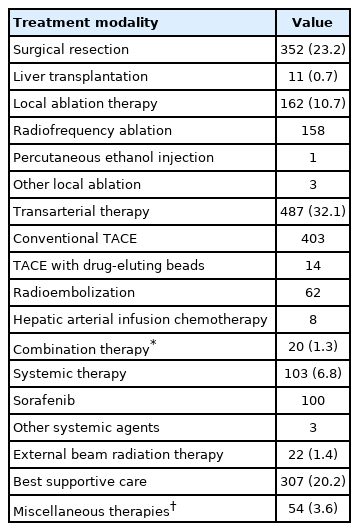
Table 2 presents the initial treatment modalities in the treatment group. Surgical treatments, including resection (23.2%) and liver transplantation (0.7%), were performed in approximately one-quarter of patients as the initial treatment. Locoregional treatments, including percutaneous ablative treatment (10.7%), external beam radiation therapy (1.4%), and transarterial treatment (32.1%), were applied in approximately 45% of patients. The vast majority of percutaneous local ablation therapies were radiofrequency ablation (97.5%). Among the transarterial therapies, 82.8% were conventional transarterial chemoembolization, 12.7% were transarterial radioembolization, and 2.9% were drug-eluting bead transarterial chemoembolization. Systemic treatment was performed in 6.8% of patients, and 20.2% underwent best supportive care only.
The treatment modalities according to BCLC stage are shown in Fig. 1. For BCLC stage 0, 61.4% of patients underwent potentially curative treatment (treatment recommended by the BCLC guidelines), including surgical resection (26.5%) or local ablation therapy (34.9%), but 31.9% of cases were treated with transarterial treatment. For BCLC stage A, 58.2% of patients were treated with surgical resection (47.0%) and local ablation therapy (11.2%), which are the recommended treatments. For BCLC stage B, 48.4% of patients were treated with transarterial treatment (recommended treatment), but 23.1% were treated with surgical resection. For BCLC stage C, only 18.1% of patients underwent systemic treatment (recommended treatment), and 30.0% were treated with transarterial treatments the initial treatment. Among BCLC stage D patients, 71.6% received best supportive care. The overall rate of adherence to the BCLC-suggested treatment was 34.5%. The rates of adherence to the BCLC treatment guidelines according to each BCLC stage are shown in Supplementary Table 1. Adherence to the BCLC guidelines was relatively high for BCLC stages 0/A (59.2%) and D (71.6%). However, for BCLC stage C, the rate of adherence to the recommended treatment (i.e., systemic treatment) was only 18.1%.

Initial treatment modality of the study population (A) and subgroups according to the Barcelona Clinic Liver Cancer (BCLC) stage 0 (B), stage A (C), stage B (D), stage C (E), and stage D (F). *Combination therapy was defined as a combined treatment with local ablation therapy and transarterial therapy; †Miscellaneous therapies were defined as unclassifiable treatment modalities (i.e., combination therapies other than transarterial therapy and local ablation therapy).
3. Overall survival
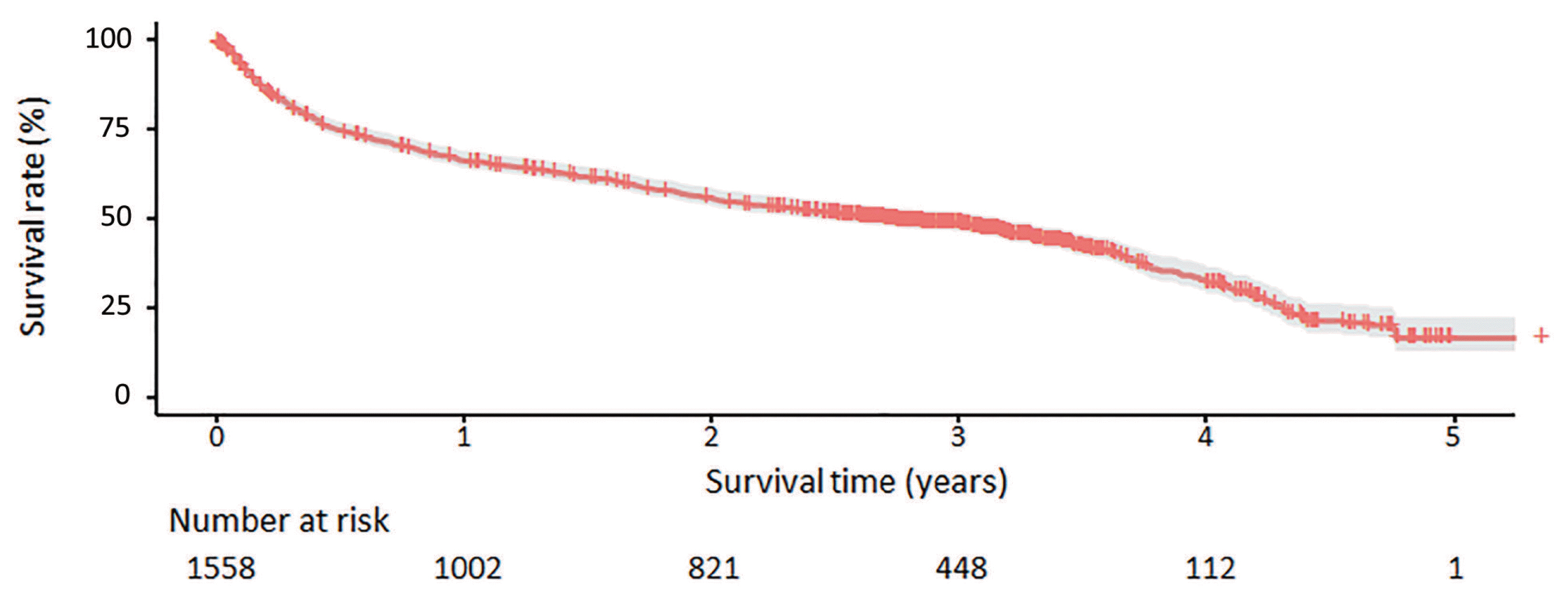
Table 3 presents the median OS and annual OS rates for the entire study population and the OS rates according to baseline characteristics. The median OS was 37.8 months (95% CI, 31.4–43.8 months), and the 1-, 3-, and 5-year OS rates were 67.1%, 50.9%, and 27.0%, respectively (Fig. 2). All survival curves showed significant survival differences according to CTP class (Fig. 3A), mUICC stage (Fig. 3B), BLCL stage after combining stages 0 and A (Fig. 3C), and AFP levels (Fig. 3D) (all log-rank test, P<0.001). The survival curves of BCLC stages 0 and A did not show any significant survival differences (log-rank test, P=0.37). We also performed post hoc analyses of log-rank tests for multiple comparisons between each survival curve and found significant survival differences between all CTP classes, between all HCC stages, and between all AFP levels (all pairwise log-rank comparisons, P<0.001). When we analyzed the median survival of patients who received initial treatment as recommended by the BCLC guidelines (Table 4), it was 59.9 months (IQR, 56.2 months-not reached) in BCLC stages 0/A, 27.0 months (IQR, 13.6–58.2 months) in stage B, 4.1 months (IQR, 1.9–7.3 months) in stage C, and 1.3 months (IQR, 0.6–3.1 months) in stage D.
Overall survival rates (%) in the study population according to baseline characteristics and initial treatment modalities
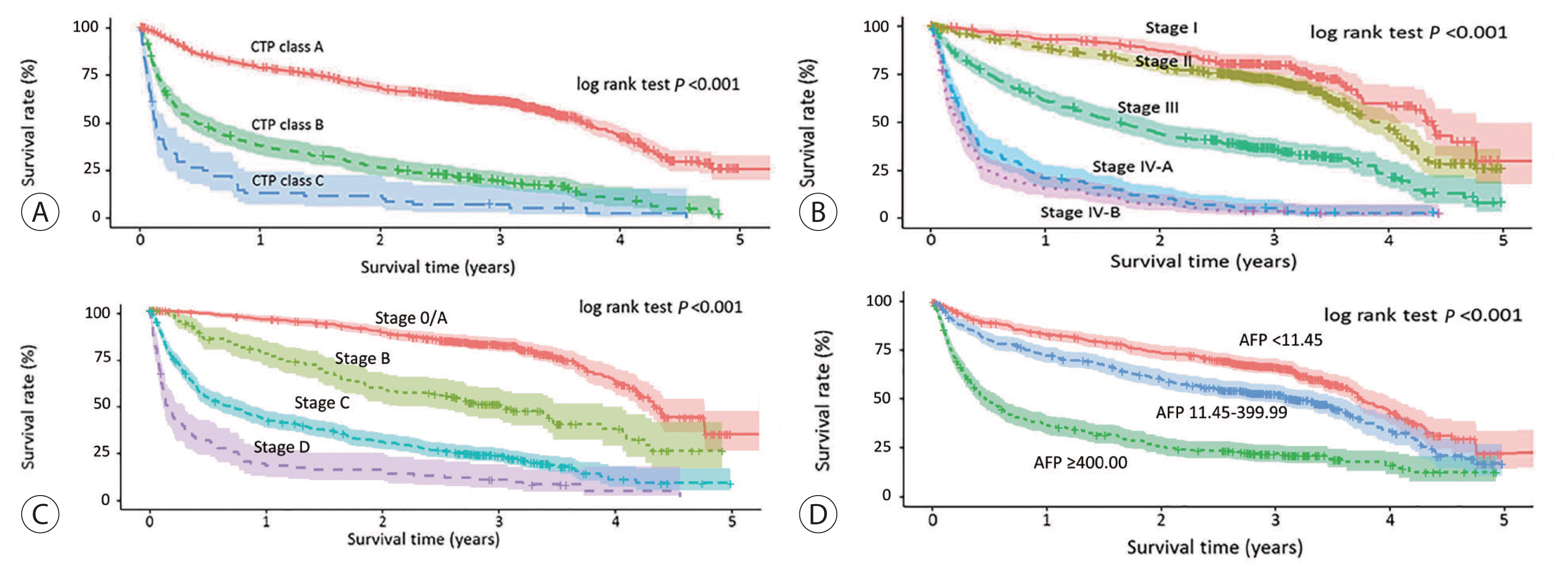
Overall survival curves for the subgroups divided by the Child–Turcotte–Pugh (CTP) classes (A), modified Union for International Cancer Control stage (B), Barcelona Clinic Liver Cancer stage (C), and serum levels of alpha-fetoprotein (AFP) (D).
DISCUSSION
The present study investigated HCC-related statistics, including baseline characteristics, treatment modalities, and survival, in Korea in 2015. We analyzed 1,558 patients with HCC registered in the KPLCR in 2015, who are representative of the general population with HCC in Korea. We showed that 45.7% of patients with HCC were diagnosed at a very early or early stage (BCLC stage 0 or A) and could be candidates for curative treatment; however, a similar proportion of patients (39%) was diagnosed at an advanced stage (BCLC stage C). Transarterial therapy (32.1%) was the most commonly performed initial treatment, followed by surgical resection (23.2%), best supportive care (20.2%), and local ablation therapy (10.7%). The 1-, 3-, and 5-year OS rates were 67.1%, 50.9%, and 27.0%, respectively. BCLC guidance for treatment was followed in 34.5% of patients. The treatment of patients with BCLC stage B and C showed relatively low adherence to this guidance.
The trends in HCC-related statistics in Korea have changed over time. We previously published reports on HCC-related statistics in Korea using data from the KPLCR between 2008 and 2011 and between 2012 and 2014.6,7 The HCC-related statistics using the data in the KPLCR between 2003 and 2005 are also available from a website established by the Korean Liver Cancer Study Group (http://www.livercancer.or.kr). Therefore, we were able to compare the HCC statistics for 2003-2005, 2008-2011, and 2012-2014 with those in 2015. We found that several characteristics, including the etiology of HCC, underlying liver function, treatment modalities, and rates of adherence to BCLC treatment guidelines, changed significantly over time. The prevalence of HBV-related HCC gradually decreased over time, while the prevalence of HCC caused by etiologies other than viral hepatitis or alcohol abuse (e.g., non-alcoholic fatty liver disease [NAFLD]) increased over time. The rates of HBV-related HCC for the periods 2003-2005, 2008-2011, 2012-2014, and 2015 were 72.3%, 72.0%, 59.1%, and 58.1%, respectively. In Korea, after the introduction of HBV vaccination in 1983 and nationwide immunization in 1995, the rate of positivity for hepatitis B surface antigen dropped from 8-10% in the 1980s to 3% in 2016.8-10 However, the prevalence of NAFLD is approximately 16-33% and is steadily increasing in Korea.11,12 Therefore, the prevalence of HCC caused by NAFLD is expected to increase in Korea. The rate of HCC caused by etiologies other than viral hepatitis or alcohol abuse increased from 5.7% in 2003-2005 to 17.0% in 2015.
The proportion of patients with preserved liver function (CTP A) increased over time. The proportion of patients with CTP A for 2003-2005, 2008-2011, 2012-2014, and 2015 were 64.2%, 71.0%, 72.9%, and 73.2%, respectively. This can be explained by advances in antiviral treatment and management of liver cirrhosis in Korea. Entecavir and tenofovir, two of the most potent antiviral agents against HBV, were introduced in 2007 and 2012, respectively.13 The application of potent antiviral agents can successfully inhibit viral replication, thereby reducing liver inflammation and fibrosis.
As the underlying liver function improved in patients with HCC over time, the proportion of patients receiving potentially curative treatment as initial treatment also increased. The proportions of patients with HCC receiving curative treatment (i.e., surgical resection, liver transplantation, and percutaneous ablative treatment) for 2003-2005, 2008-2011, 2012-2014, and 2015 were 17.1%, 28.2%, 31.5%, and 34.6%, respectively. The rate of patients receiving systemic treatment also increased from 0.8% in 2003-2005 to 6.8% in 2015. Considering that approximately 40% of HCC cases are identified at an advanced stage and that novel systemic agents for first-line treatment (e.g., lenvatinib and atezolizumab plus bevacizumab) and second-line or later treatment (e.g., regorafenib, nivolumab, ramucirumab, pembrolizumab, and cabozantinib) have recently been introduced14-20, systemic treatment is expected to become more widely used for patients with HCC in the near future. In contrast, although transarterial treatment was the most commonly used treatment modality in all periods, the proportion of patients with HCC receiving transarterial treatment as the initial treatment has been gradually decreasing over time (49.5%, 41.7%, 37.5%, and 32.1% for the periods 2003-2005, 2008-2011, 2012-2014, and 2015, respectively).
The overall adherence rates for the periods 2008-2011, 2012-2014, and 2015 were 37.7%, 38.2%, and 34.5%, respectively. As the proportion of patients with HCC receiving curative treatment has increased over time, the adherence rate of patients at BCLC stage 0/A also increased (47.4%, 54.8%, and 59.2% for the periods 2008-2011, 2012-2014, and 2015, respectively). The adherence rate of patients at BCLC stage C has also increased as the number of HCC patients receiving systemic treatment has increased (9.5%, 11.7%, and 18.1% for the periods 2008-2011, 2012-2014, and 2015, respectively). However, unexpectedly, the adherence rate of patients at BCLC stage B decreased over time (62.8%, 55.1%, and 48.4% for the periods 2008-2011, 2012-2014, and 2015, respectively). This trend might be due to an increase in the proportion of patients with HCC at BCLC stage B who underwent surgical resection (10.2%, 21.1%, and 23.1% for the periods 2008-2011, 2012-2014, and 2015, respectively). A previous meta-analysis indicated that surgical resection provides a significant survival benefit over transarterial treatment in patients with BCLC stage B HCC when the HCC is resectable.21 Because HCCs at BCLC stage B are highly heterogeneous, more stratified and individualized treatment guidelines are warranted, considering liver function, tumor extent, and the level of vascular invasion.
In conclusion, we have shown that in 2015, approximately 45% of HCCs were diagnosed at a very early or early stage, and 35% of patients received potentially curative treatment as their initial treatment. The BCLC guidelines for treatment were followed in 34.5% of patients with HCC; there was relatively low adherence to these guidelines in patients with BCLC stage B or C HCC. We believe that the results and highlighted trends in the characteristics of HCC in Korea will contribute to improve the management of this disease and promote further study.
Supplementary Data
Flow diagram of the study population. KPLCR, Korean Primary Liver Cancer Registry; HCC, hepatocellular carcinoma.
Rates of adherence to the BCLC treatment guideline in each BCLC stage and the total study population
Notes
AUTHORSHIP STATEMENT
1) Jeong-Hoon Lee takes responsibility for the integrity of the work as a whole, from inception to published article.
-
2) Specific author contributions
Conception: Jun Sik Yoon, Han Ah Lee, and Jeong-Hoon Lee; study design: Jun Sik Yoon, Han Ah Lee, and Jeong- Hoon Lee; participation in patient management and data collection: Jun Sik Yoon, Han Ah Lee, Hwi Young Kim, Dong Hyun Sinn, Dong Ho Lee, Ju-Yeon Cho, Jonggi Choi, Young Chang, Suk Kyun Hong, Hyun-Joo Kong, Young-Joo Won, Eunyang Kim, and Jeong-Hoon Lee; contribution to the data acquisition, responsibility for writing the paper, and statistical analysis: Jun Sik Yoon, Han Ah Lee, and Jeong-Hoon Lee
3) All authors have reviewed the paper and approved the final version.
Conflicts of Interest
The authors have no financial conflicts of interest.
Acknowledgements
The database used in this study was provided by the Korean Central Cancer Registry, Ministry of Health and Welfare, South Korea, and the Korean Liver Cancer Association.