Articles
- Page Path
- HOME > J Liver Cancer > Volume 22(2); 2022 > Article
-
Case Report
Hepatocellular carcinoma with Budd-Chiari syndrome due to membranous obstruction of the inferior vena cava with long-term follow-up: a case report -
Choong Hee Kim1
 , Gwang Hyeon Choi1
, Gwang Hyeon Choi1 , Hee Young Na2
, Hee Young Na2 , Chang Jin Yoon3
, Chang Jin Yoon3 , Jai Young Cho4
, Jai Young Cho4 , Sangmi Jang1
, Sangmi Jang1 , Ji Hye Kim1
, Ji Hye Kim1 , Eun Sun Jang1
, Eun Sun Jang1 , Jin-Wook Kim1
, Jin-Wook Kim1 , Sook-Hyang Jeong1
, Sook-Hyang Jeong1
-
Journal of Liver Cancer 2022;22(2):194-201.
DOI: https://doi.org/10.17998/jlc.2022.08.24
Published online: September 15, 2022
1Department of Internal Medicine, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Korea
2Department of Pathology, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Korea
3Department of Radiology, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Korea
4Department of General Surgery, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Korea
-
Corresponding author: Gwang Hyeon Choi Department of Internal Medicine, Seoul National University Bundang Hospital, Seoul National University College of Medicine, 82 Gumi-ro 173beon-gil, Bundang-gu, Seongnam 13620, Korea
Tel. +82-31-787-7029, Fax. +82-31-787-4052 E-mail: gwanghyeon@nate.com
Copyright © 2022 The Korean Liver Cancer Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 2,299 Views
- 55 Downloads
Abstract
- Membranous obstruction of the inferior vena cava (MOVC) is a rare subset of Budd-Chiari syndrome (BCS) with a subacute onset that is often complicated by cirrhosis and hepatocellular carcinoma (HCC). Here we report a case of recurrent HCC in a patient with cirrhosis and BCS that was treated with several episodes of transarterial chemoembolization followed by surgical tumorectomy, whereas the MOVC was successfully treated with balloon angioplasty followed by endovascular stenting. The patient was followed up for 9.9 years without anticoagulation and experienced no stent thrombosis. After the tumorectomy, the patient was HCC-free for 4.4 years of follow-up.
- Budd-Chiari syndrome (BCS) is obstruction of the hepatic venous outflow from the small hepatic venules to the entrance of the inferior vena cava (IVC) into the right atrium (RA) regardless of its etiology.1,2 BCS induces chronic liver congestion, causing hepatomegaly, ascites, leg edema, collateral venous dilatation in the trunk, and portal hypertension.3 Previous studies suggested that hepatic congestion caused by obstruction of hepatic venous outflow can be complicated by cirrhosis and subsequent hepatocellular carcinoma (HCC).4,5
- Membranous obstruction of the IVC (MOVC), whether partial or complete, with or without hepatic vein involvement, is a rare but potentially treatable subset of BCS. In contrast to hepatic venous outflow tract obstruction in classical BCS, which involves hepatic vein thrombosis, MOVC involves mechanical obstruction of the hepatic portion of the IVC just below the diaphragm. MOVC predominantly occurs in Asian and African populations, whereas classical BCS mainly occurs in United States and European Caucasian populations.6 Thus, although both MOVC and classical BCS are caused by hepatic venous outflow obstructions, their pathophysiologies, clinical presentations, and clinical courses differ.5,7,8
- Here, we report a case of recurrent HCC caused by MOVC in a patient with cirrhosis and BCS caused by MOVC. The recurrent HCC was treated with several episodes of transarterial chemoembolization (TACE) followed by surgical tumorectomy, while the MOVC was successfully treated with balloon angioplasty followed by endovascular stenting. This case report is written in accordance with the CARE guidelines (available at https://www.care-statement.org/).
INTRODUCTION
- A 66-year-old woman presented to the outpatient clinic of Seoul National University Bundang Hospital with cirrhosis of the liver and a liver nodule detected on upper abdomen ultrasonography during a health check-up. The patient claimed to have received a transfusion for peritonitis approximately 35 years prior. She did not take any medications on a regular basis, worked as a housewife, was a non-smoker, and drank 1-2 bottles of alcohol per month. The patient denied a family history of liver disease. The patient was 172 cm tall, weighed 46 kg, and had a body mass index of 15.5. On presentation, the patient was asymptomatic, and a review of the system and physical examination findings was unremarkable. The patient showed no blatant stigmata of chronic liver disease such as abdominal distention or spider angioma.
- Initial lab findings showed the following: white blood cell count, 4,630/mm3; hemoglobin concentration, 14.9 g/dL; platelet count, 101,000/mm3; serum albumin, 4.5 g/dL; total bilirubin, 1.8 mg/dL; direct bilirubin, 0.3 mg/dL; aspartate aminotransferase to alanine aminotransferase (AST/ALT), 15/11 IU/L; alkaline phosphatase, 100 IU/L; γ-glutamyl transferase, 18 IU/L; and prothrombin time international normalized ratio, 1.09. The patient’s tumor markers showed a mildly elevated prothrombin induced by vitamin K absence-II (PIVKA-II) value of 50 mAU/mL and a normal serum alpha-fetoprotein (AFP) value of 7.9 ng/mL. The patient tested negative for viral markers, including hepatitis B surface antigen, hepatitis B surface antibody, and hepatitis C antibody; hepatitis B core antigen was not ordered in the baseline evaluation. The patient showed normal ranges for protein C and S, and was negative for anti-nuclear antibody, lupus anticoagulant, anti-cardiolipin immunoglobin M and G, rheumatoid factor, human immunodeficiency virus antibody, and non-reactive venereal disease research laboratory tests.
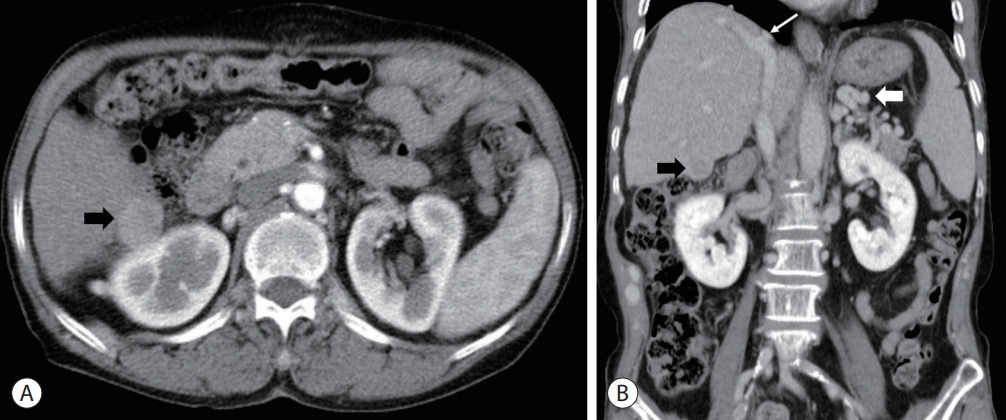
- Initial multiphase detector liver computed tomography (CT) imaging showed cirrhotic changes in the liver contour with splenomegaly, esophageal varices, scanty ascites in the pelvic cavity, a single 2.5 cm arterialphase enhancing, delayed-phase low-attenuating exophytic nodular mass in segment 6 (S6) of the right hepatic lobe suggestive of HCC, and obstruction of the intrahepatic portion of the IVC with mural calcification along the hepatic dome area suggestive of MOVC (Fig. 1). Although the right hepatic vein drained into the IVC, the proximal segment of the middle and left hepatic veins was obstructed by thrombosis or fibrotic changes and drained into the right hepatic vein via its collateral branches. In addition, vascular structures linking segment 3 with the portal vein as well as several prominent collateral veins along the abdominal wall and vertebrae were noted, suggesting the presence of portosystemic collateral circulation.
- The patient underwent inferior vena cavography using the right femoral vein approach. Preangioplasty venography showed focal complete obstruction at the IVC and RA junction, compatible with a diagnosis of MOVC (Fig. 2), and venous blood draining into the RA through the paravertebral collateral veins. After confirming the diagnosis of MOVC using IVC venography, the obstruction was punctured with a metallic needle via the right internal jugular vein approach. A guidewire was then passed through the obstruction site, and balloon angioplasty was performed, followed by the insertion of a self-expandable stent (14 mm×6 cm). Post-angioplasty venography showed patent venous flow through the fully expanded stent and decreased collateral venous flow. The patient simultaneously underwent TACE for HCC in hepatic S6. The patient experienced no post-procedural complications and was discharged without anticoagulants or antiplatelet agents.
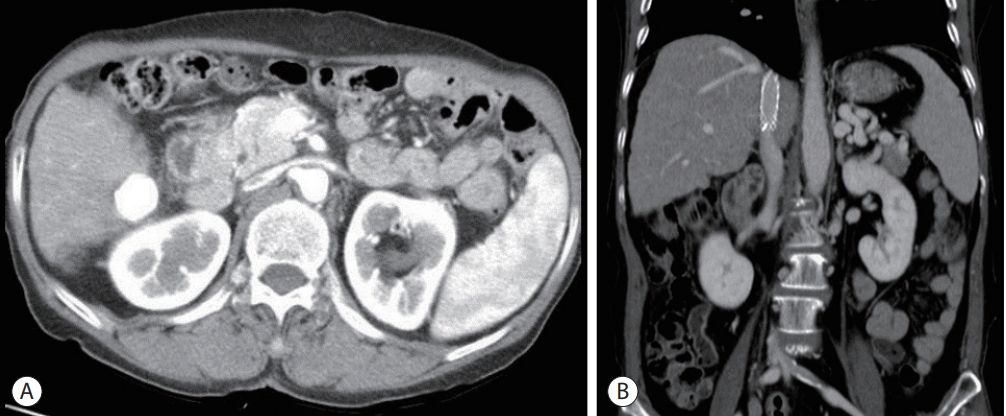
- One month after TACE and IVC stenting, an interval liver-protocol CT scan showed compact lipiodol uptake in the HCC of S6 and no evidence of residual viable HCC (Fig. 3). The inferior IVC stent was patent. The AFP level had decreased from 7.9 ng/mL to 4.1 ng/mL, while the PIVKA-II had decreased from 50 mAU/mL to 15 mAU/mL. Thereafter, the patient visited the outpatient clinic every 3-6 months for a liver-protocol CT scan and AFP and PIVKA-II testing.
- Three years after the initial diagnosis, liver-protocol CT imaging showed a patent IVC stent; however, a 1.9 cm exophytic recurrent HCC at segment 6 adjacent to the previously treated lesion was noted. Furthermore, the PIVKA-II level had increased from 15 mAU/mL to 105 mAU/mL, although AFP changed little from 4.1 ng/mL to 4.0 ng/mL. Although surgery was initially suggested, the patient was reluctant to accept it; furthermore, since the response to the initial TACE was satisfactory, the patient underwent additional TACE rather than surgery. Following the second and third TACE sessions, 1-monthinterval liver CT imaging showed no residual viable HCC with a patent IVC stent. Moreover, the PIVKA-II level decreased from 105 mAU/mL to 16 mAU/mL (normal) and the AFP remained stable, changing from 4.0 ng/mL to 3.9 ng/mL.
- Four years and 4 months after the initial diagnosis, liverprotocol CT imaging revealed a recurrent 2.1 cm nodular lesion with arterial enhancement and delayed washout in S6 near the previous TACE site. Tumor makers were mildly increased, with the AFP changing from 3.9 ng/mL to 4.8 ng/mL and PIVKA-II changing from 16 mAU/mL to 20 mAU/mL. The patient was readmitted, and a fourth session of TACE was performed on the recurrent mass. One-month-interval liver CT imaging showed no evidence of residual viable HCC.
- Five years after the initial diagnosis, liver-protocol CT imaging showed a newly developed, 1-cm arterial enhancing lesion without definite washout close to the previously treated S6 lesion suggestive of an indeterminate nodule. Tumor marker levels were unremarkable, with AFP changing from 3.3 ng/mL to 3.9 ng/mL and no change in the PIVKA-II values (16 mAU/mL). The patient underwent a fifth TACE session; however, 1 month-interval liver CT imaging still showed residual HCC around the previously treated lesion. Laboratory tests showed a platelet count of 101,000/mm3, serum albumin of 4.5 g/dL, and total bilirubin of 1.8 mg/dL. At this point, it was evident that the recurrent HCC lesion was refractory to TACE. After obtaining patient consent, our team planned surgical resection. In retrospect, resection could have been considered at an early stage.
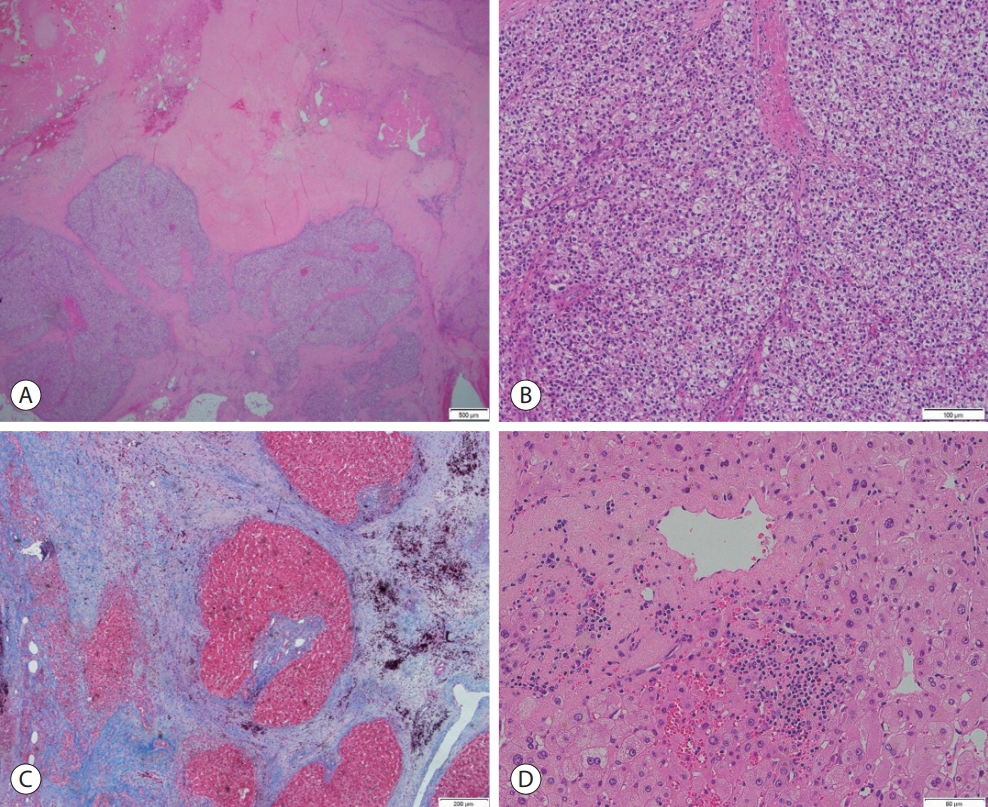
- Five years and 4 months after the initial diagnosis, the patient underwent S6 tumorectomy with cholecystectomy. A histopathological examination of the tumorectomy specimen showed two masses: one was a 4.0 cm completely necrotic mass, while the other was a 1.3 cm HCC with 40% tumor necrosis and 50% hemorrhage, predominantly grade II differentiation without major vessel or bile duct invasion but with capsular infiltration and peritoneal invasion and a negative resection margin. Thus, the final pathologic staging of ypT4Nx was made (Fig. 4A, B). Non-tumor pathology of the liver showed macro- and micronodular cirrhosis with reverse lobulation (venocentric) pattern, septal hyalinization and hemorrhage, minimal lobular activity, minimal septal activity, and macro- and microvesicular steatosis grade 1+ of 3 (Fig. 4C, D). The sinusoidal spaces showed mild sinusoidal dilatation with congestion, a finding compatible with BCS. Postoperatively, the patient was discharged without complications, and one-month-interval CT imaging showed no residual tumor in the abdomen.
- Nine years and 11 months after the initial diagnosis (4 years and 6 months after surgical tumorectomy, respectively), a liver-protocol CT scan showed no evidence of HCC recurrence or IVC stent complications. A review of all previous liver-protocol CT scans over a 10-year observation period showed no significant shrinkage or change in the size of collateral vessels or splenomegaly. Tumor markers were stable after resection, and the last follow-up values were an AFP of 4.6 ng/mL and a PIVKA-II of 11 mAU/mL. The patient was eventually referred in stable condition to a locoregional center because of the proximity of care and her personal preference.
CASE REPORT
- This was a case of a 66-year-old woman with no underlying disease or clinical symptoms who was referred to a tertiary medical center due to an incidental liver space-occupying lesion found during screening ultrasonography. Imaging studies have shown the presence of MOVC and HCC in liver cirrhosis. MOVC was confirmed by inferior vena cavography, and the lesion was subsequently treated with balloon angioplasty followed by stent insertion. Although the HCC was initially treated with TACE, repeated local recurrence led to additional TACE, and the patient ultimately underwent surgical tumorectomy at the recurrent HCC site. The unique feature of this case is that the patient was followed up for 9.9 years after endovascular treatment without anticoagulation, during which time no stent thrombosis or other stent-related complications were noted. In addition, other than the recurrent HCC in S6, which was treated with local therapy in the form of TACE and surgical tumorectomy, no additional HCC developed in other liver segments. Following the tumorectomy of S6, the patient remained HCC-free for the remainder of the follow-up period.
- In BCS due to MOVC, the culprit obstructive structure within the hepatic portion of the IVC may appear as a thin membrane, typically between the connection of the right and middle hepatic veins to the IVC (type I), or a fibrous cord of various lengths (type II).9 Although its pathogenesis is largely unknown, Okuda10 suggested that the organization of repeated thrombosis along the hepatic portion of the IVC, rather than a congenital vascular malformation, may cause the fibrous structure. How, why, and the circumstances under which the membranous structure forms along the hepatic portion of the IVC remains unclear.
- The clinical presentation of BCS due to MOVC is usually chronic and insidious, with long asymptomatic periods interrupted by recurrent acute exacerbations.8 Due to pooled blood in the IVC flowing back into the hemiazygos and azygos veins and then into the iliolumbar veins, the azygos and hemiazygos veins may be dilated in imaging studies. However, if these deep collateral venous channels are not well-developed, prominent collateral veins in the cephalad direction may be visualized in the anterior and posterior regions of the trunk.11 Lower-extremity edema with or without venous stasis may be present. The liver is usually firm but not acutely enlarged or tender. Ascites may or may not be observed. The most frequent clinical symptom is intermittent or continuous abdominal discomfort followed by intermittent or continuous ankle swelling.8,11 Due to the chronic nature of BCS due to MOVC, the liver often becomes cirrhotic and may be complicated by HCC; a review of biopsied cases by Shrestha found that 70% of patients with BCS due to MOVC developed liver cirrhosis and more than 20% developed HCC.8
- Biopsy findings of BCS with MOVC may range from normal or minimal changes in congestive hepatopathy, such as sinusoidal dilatation or central vein dilatation, to advanced features of congestive hepatopathy, including congestion, coagulative necrosis, or loss of predominantly centrilobular hepatocytes without inflammatory infiltrates and/or fibrosis.7,8 These pathological findings are not exclusive to BCS with MOVC; rather, they are also present in veno-occlusive disease or secondary hepatopathy due to cardiac or pericardial conditions. The patient in this study showed mild sinusoidal dilatation on pathological examination despite the presence of liver cirrhosis due to MOVC, possibly because the surgical specimen was obtained 5 years after portal decompression with IVC stent insertion, resulting in improved sinusoidal dilatation.
- Percutaneous transluminal angioplasty is usually effective in MOVC. One third of patients with MOVC have shortlength stenosis of the hepatic veins or IVC, making them ideal candidates for percutaneous transluminal angioplasty.7 The retrograde, transjugular, and transfemoral routes are all safe and efficacious. Treatment modalities include transcardiac membranotomy, vena caval-atrial bypass surgery, shunt surgery, balloon angioplasty, and expandable stent insertion.7 Wu et al. reported that 97.6% of patients with MOVC were successfully treated with balloon angioplasty, while Valla reported a success rate of over 96% for lesions in the IVC, with 5-year cumulative primary and secondary patency rates of 80% and 90%, respectively.11,12 A retrospective review of three tertiary hospitals in Korea reported that, among 17 patients with MOVC who underwent angioplasty and were followed for an average of 4.5 years, the primary patency rate was 76.5%, the primary assisted patency rate was 94.1%, and no deaths were noted during the follow-up period.13
- The role of anticoagulation therapy following angioplasty for MOVC has not been well defined. Valla suggested anticoagulation therapy for all patients regardless of their prothrombotic disorder status; however, the study did not make a clear distinction between classical BCS due to hepatic vein thrombosis and BCS due to MOVC.11 A review by Shrestha reported that neither thrombolytic therapy nor anticoagulation therapy effectively treated MOVC, yet the study did not specify angioplasty status.8 According to Lee et al.13, all 14 patients who underwent endovascular procedures were on antiplatelet treatment for 1 month after the procedures; after 2 years of follow-up, the restenosis rate was 23.5%.
- Despite the excellent patency rates following angioplasty, the review by Shrestha found that surgical or endovascular procedures for the treatment of MOVC did not significantly prevent the development of cirrhosis or HCC. Instead, it showed that the development of cirrhosis in MOVC was significantly related to the severity and frequency of acute exacerbations.8 Takamura et al.14 reported a case of HCC recurrence at 102 months after successful percutaneous transluminal angioplasty and hepatic resection for HCC.
- The development of HCC is well documented in BCS with MOVC. Studies in South African Blacks showed a prevalence of HCC of 40-48% in patients with MOVC, while a study in Japan showed a prevalence of 41%.7 A small cohort study of 67 patients with BCS due to MOVC in Korea showed a cumulative incidence of 42.6% after 15 years with an annual incidence of 2.8%,11 while a retrospective review of three teaching hospitals in Korea over a 5-year study period showed the development of HCC in four of 28 patients with BCS due to MOVC for a prevalence of 14.3%.13 MOVC-associated HCC appears to differ from HCC in HBV infection by a uni- or pauci-nodular pattern, peripheral location, well differentiation, less frequent portal venous invasion, and overall less aggressive nature.11 TACE appears to be well tolerated and may be associated with prolonged survival in patients with BCS. A retrospective review of 23 patients with MOVC-associated HCC who underwent TACE showed an incidence of HCC in MOVC diagnosed patients of 23% (23 of 98 patients), mean age of 48.7 years at diagnosis, 61% complete response rate, 17% partial response rate, and 30% recurrence rate at a median 15.7-month interval. A significant decrease in AFP level following TACE (P<0.001) and a significant increase in AFP level after recurrence were observed (P=0.018). One- and 5-year survival rates on Kaplan-Meier analysis were 90% and 46%, respectively. A The peripheral distribution of MOVC-associated HCC was also observed in this study.15
- In conclusion, this case report presented several key features of BCS for MOVC complicated by HCC. Despite requiring repeated local therapies for recurrent HCC, the patient showed excellent long-term IVC stent patency following angioplasty and favorable outcomes.
DISCUSSION
-
Conflicts of Interest
The authors have no conflicts of interest to disclose.
-
Ethics Statement
The Institutional Review Board (IRB) of Seoul National University Bundang Hospital approved this study (IRB No. B-2208-774-702) and waived the requirement for informed consent.
-
Funding Statement
No funding to declare.
-
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed for this case report.
-
Author Contribution
Conceptualization: GHC, SHJ
Data curation: GHC, SHJ, HYN
Formal analysis: GHC
Funding acquisition: none
Investigation: GHC
Methodology: GHC
Project administration: GHC
Resources: GHC
Software: CHK
Supervision: SHJ
Validation: SHJ
Visualization: CHK, GHC
Writing–original draft: CHK
Writing–review & editing: CHK, GHC, SJ
Approval of final manuscript: all authors
Article information

- 1. Ludwig J, Hashimoto E, McGill DB, van Heerden JA. Classification of hepatic venous outflow obstruction: ambiguous terminology of the Budd-Chiari syndrome. Mayo Clin Proc 1990;65:51−55.ArticlePubMed
- 2. Shukla A, Shreshtha A, Mukund A, Bihari C, Eapen CE, Han G, et al. Budd-Chiari syndrome: consensus guidance of the Asian Pacific Association for the study of the liver (APASL). Hepatol Int 2021;15:531−567.ArticlePubMedPDF
- 3. Matsui S, Ichida T, Watanabe M, Sugitani S, Suda T, Takahashi T, et al. Clinical features and etiology of hepatocellular carcinoma arising in patients with membranous obstruction of the inferior vena cava: in reference to hepatitis viral infection. J Gastroenterol Hepatol 2000;15:1205−1211.ArticlePubMed
- 4. Okuda K. Inferior vena cava thrombosis at its hepatic portion (obliterative hepatocavopathy). Semin Liver Dis 2002;22:15−26.Article
- 5. Okuda K, Kage M, Shrestha SM. Proposal of a new nomenclature for Budd-Chiari syndrome: hepatic vein thrombosis versus thrombosis of the inferior vena cava at its hepatic portion. Hepatology 1998;28:1191−1198.ArticlePubMed
- 6. Simson IW. Membranous obstruction of the inferior vena cava and hepatocellular carcinoma in South Africa. Gastroenterology 1982;82:171−178.ArticlePubMed
- 7. Kew MC, Hodkinson HJ. Membranous obstruction of the inferior vena cava and its causal relation to hepatocellular carcinoma. Liver Int 2006;26:1−7.ArticlePubMedPDF
- 8. Shrestha SM. Liver cirrhosis in hepatic vena cava syndrome (or membranous obstruction of inferior vena cava). World J Hepatol 2015;7:874−884.ArticlePubMedPMC
- 9. Lim JH, Park JH, Auh YH. Membranous obstruction of the inferior vena cava: comparison of findings at sonography, CT, and venography. AJR Am J Roentgenol 1992;159:515−520.ArticlePubMed
- 10. Okuda K. Membranous obstruction of the inferior vena cava (obliterative hepatocavopathy, Okuda). J Gastroenterol Hepatol 2001;16:1179−1183.ArticlePubMed
- 11. Valla DC. Budd-Chiari syndrome/hepatic venous outflow tract obstruction. Hepatol Int 2018;12(Suppl 1): 168−180.ArticlePubMedPDF
- 12. Wu T, Wang L, Xiao Q, Wang B, Li S, Li X, et al. Percutaneous balloon angioplasty of inferior vena cava in Budd-Chiari syndromeR1. Int J Cardiol 2002;83:175−178.ArticlePubMed
- 13. Lee BB, Villavicencio L, Kim YW, Do YS, Koh KC, Lim HK, et al. Primary Budd-Chiari syndrome: outcome of endovascular management for suprahepatic venous obstruction. J Vasc Surg 2006;43:101−108.ArticlePubMed
- 14. Takamura M, Ichida T, Yokoyama J, Matsuda Y, Nomoto M, Aoyagi Y. Recurrence of hepatocellular carcinoma 102 months after successful eradication and removal of membranous obstruction of the inferior vena cava. J Gastroenterol 2004;39:681−684.ArticlePubMedPDF
- 15. Gwon D 2nd, Ko GY, Yoon HK, Sung KB, Kim JH, Lee SS, et al. Hepatocellular carcinoma associated with membranous obstruction of the inferior vena cava: incidence, characteristics, and risk factors and clinical efficacy of TACE. Radiology 2010;254:617−626.ArticlePubMed
References
Figure & Data
References
Citations


 E-submission
E-submission THE KOREAN LIVER CANCER ASSOCIATION
THE KOREAN LIVER CANCER ASSOCIATION
 PubReader
PubReader ePub Link
ePub Link Download Citation
Download Citation

 Follow JLC on Twitter
Follow JLC on Twitter