Liver transplantation for hepatocellular carcinoma with portal vein tumor thrombosis
Article information
Abstract
Traditionally, liver transplantation for hepatocellular carcinoma with portal vein tumor thrombosis is not recommended. However, with recent developments in locoregional therapies for hepatocellular carcinoma, more aggressive treatments have been attempted for advanced hepatocellular carcinoma. Recently, various studies on locoregional therapies for downstaging followed by living donor liver transplantation reported inspiring overall survival and recurrence-free survival of patients. These downstaging procedures included three-dimensional conformal radiation therapy, trans-arterial chemoembolization, stereotactic body radiation therapy, trans-arterial radioembolization, hepatic arterial infusion chemotherapy and combinations of these therapies. Selection of the optimal downstaging protocol should depend on tumor location, biology and background liver status. The risk factors affecting outcome include pre-downstaging alpha-fetoprotein values, delta alpha-fetoprotein values, disappearance of portal vein tumor thrombosis on imaging and meeting the Milan criteria or not after downstaging. For hepatocellular carcinoma with portal vein tumor thrombosis, downstaging procedure with liver transplantation in mind would be helpful. If the reaction of the downstaged tumor is good, liver transplantation may be performed.
INTRODUCTION
Liver transplantation (LT) is an effective treatment for nonresectable hepatocellular carcinoma (HCC) with liver cirrhosis. However, LT requires adequate conditions to be successful, including tumor size, tumor number, vascular invasion, and extrahepatic metastasis. The Milan criteria (1996) and University of California San Francisco criteria (UCSF, 2001) have been generally used for selection; while these two criteria conflict regarding ideal tumor size, they agree that the absence of vascular invasion is an indication for LT.1,2 Vascular invasion is a poor prognostic factor for HCC, resulting in a median survival time of two to 12 months, and is regarded as a contraindication for LT.3 Sorafenib is the only recommended treatment for advanced HCC according to the Barcelona Clinic Liver Cancer (BCLC) staging system. However, the outcome of sorafenib-alone treatment is still poor, while advanced HCC is relatively common with a 10% to 40% prevalence rate in total HCC population.4,5 Thus, medical centers are expanding their treatment profile for advanced HCC, many of which have been well-reviewed in a recent article.6 As locoregional therapy develops and good outcomes are achieved with advanced HCC, an increasing variety of downstaging procedures for advanced HCC followed by LT are being performed. We seek herein to review living donor LT (LDLT) outcomes after downstaging for HCC with portal vein tumor thrombosis (PVTT).
HCC WITH PVTT DIAGNOSIS AND STAGING
Ultrasound (US), contrast-enhanced US (CEUS), computed tomography (CT), and magnetic resonance imaging (MRI) are generally used as diagnostic methods for HCC with PVTT. CEUS is a cheap and highly sensitive method for differential diagnosis of malignant PVTT and benign lesions. 7 Malignant PVTT shows increased enhancement in the arterial phase and rapid wash-out in the portal/delayed phase, similar to HCC.8 CT imaging and MRI are usually used for diagnosing liver status or collateral vessels and for HCC evaluation and may also be useful for differentiating benign lesions and malignant PVTT with a sensitivity and specificity of around 90%.9,10 18-fluorodeoxyglucose positron emission tomography (PET) can reveal the metabolic activity of malignant PVTT. One study showed that PET/CT can differentiate between benign lesions and malignant PVTT with a sensitivity of 93.6% and a specificity of 80% when the criterion of maximum standardized uptake value (SUVmax) was set to more than 3.35.11
There are some studies that have classified the grade of PVTT in many ways. One of the well-known classifications is from the Liver Cancer Study Group of Japan.12 Under this scheme, a PVTT case is distinguished as one of the following four grades: Vp1, tumor thrombus distal to the second-order branch of the portal vein but not in the second-order branch; Vp2, tumor invasion of the second-order branch; Vp3, tumor invasion of the first-order branch; and Vp4, tumor invasion of the main portal vein (MPV) and/or the portal vein branch contralateral to the primarily involved lobe (Fig. 1).
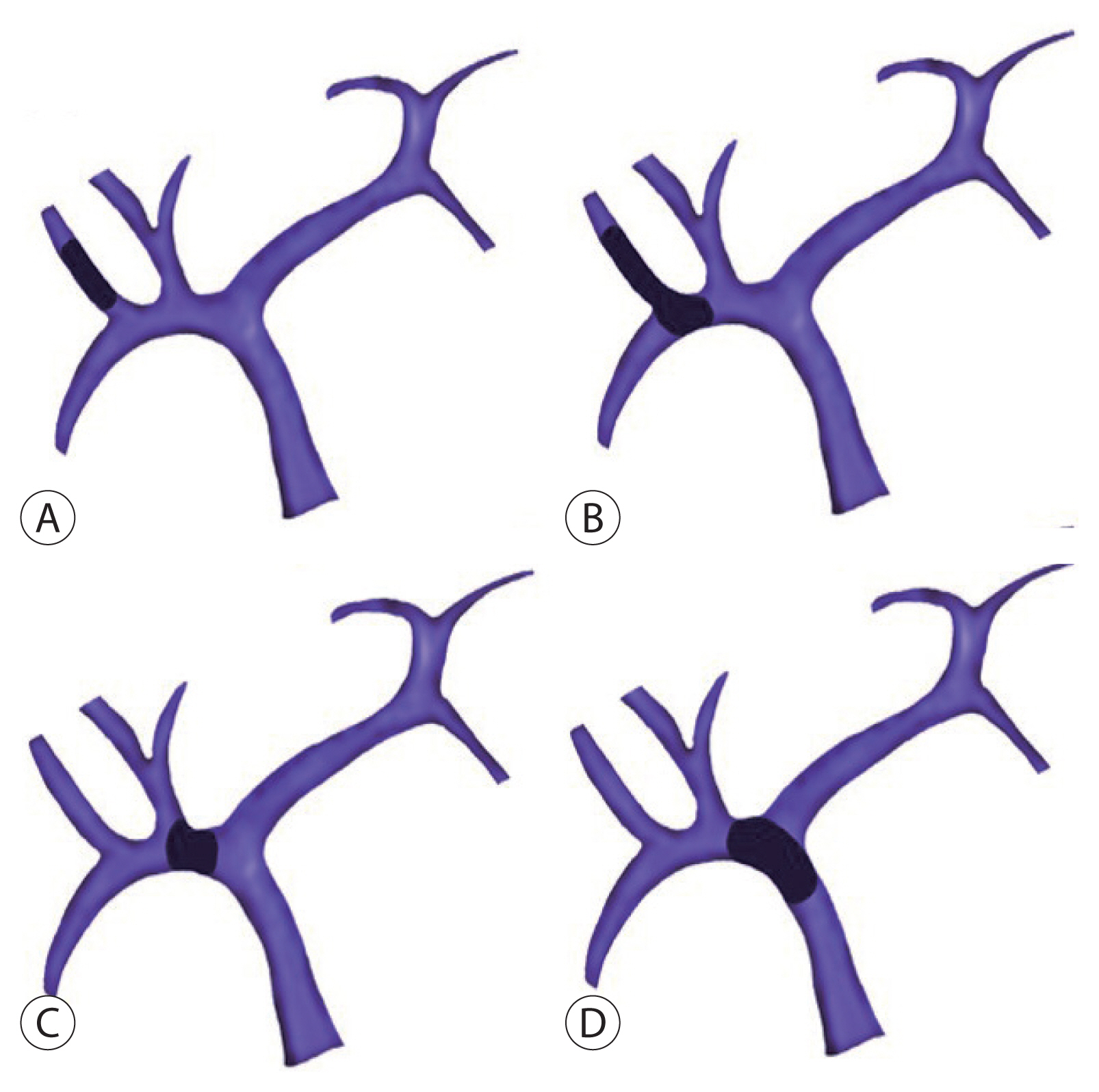
Portal vein tumor thrombosis classification according to location. (A) Vp1, tumor thrombus distal to the second-order branch of the portal vein but not in the second-order branch. (B) Vp2, tumor invasion of the second-order branch. (C) Vp3, tumor invasion of the first-order branch. (D) Vp4, tumor invasion of the main portal vein and/or the portal vein branch contralateral to the primarily involved lobe.
NONSURGICAL THERAPEUTIC OPTIONS FOR HCC WITH PVTT
Sorafenib, a multi-targeting tyrosine kinase inhibitor, is recommended for the treatment for HCC with PVTT according to BCLC staging.13 This drug supported relatively longer survival rates relative to the placebo in two phase III trials.14,15 Also, the combination of sorafenib and locoregional therapies has been trialed; here, sorafenib combined with transarterial chemoembolization (TACE) or sorafenib combined with radiofrequency ablation (RFA) for HCC with PVTT led to relatively better outcomes than sorafenib-alone treatment.16,17 However, these combination therapies possess a risk for high adverse events such as abdominal pain, hand-foot skin reactions, asthenia, diarrhea, and weight loss.
TACE, which has been accepted as palliative therapy for multinodular HCC in a still-functioning liver, has a risk for inducing ischemia of the normal liver when insufficient collateral vessels exist around a tumor-obstructed portal vein.6,18 However, in selective cases, TACE achieved better outcomes than those of the supportive group in the setting of HCC with PVTT. A recent study reviewed 1,933 patients who received TACE for HCC with PVTT and reported one-, three-, and five-year overall survival (OS) rates of 29%, 4%, and 1%, respectively.19 Sorafenib, which expresses anti-angiogenic activity—which may suppress the angiogenic factor released due to hypoxia after TACE—can be combined with TACE, resulting in better OS rates.20 There is also a recently inspiring result of combined TACE and sorafenib with a three-year OS of 86.1% in a phase II trial study.21
Steady efforts to explore the impact of radiation therapy (RT) on HCC with PVTT revealed some effects even despite the risk of radiation-induced liver disease (RILD). However, one study reported that a high dose of radiation applied to a large liver volume (V30Gy >28.1%) may cause RILD, resulting in jaundice, hepatomegaly, ascites, and an elevated Child-Pugh score.22 To avoid injury to nontumorous areas of the liver, three-dimensional conformal RT (3D-CRT) was developed, which applies radiation highly selectively to the tumor and PVTT by modifying the radiation beam to match the shape of the tumor.23 Also, the use of a “dose-volume histogram” may guide radiation concentrated on the tumor.24 A recent propensity score-matching study showed that RT resulted in impressively longer median survival times as compared with sorafenib treatment in 56 of Vp3 or Vp4 PVTT patients (10.9 months vs. 4.8 months).25 Other investigations of stereotactic body RT (SBRT), proton beam therapy, and gamma knife radiosurgery showed comparable outcomes in HCC with PVTT.26,27
Although there is a risk of complications such as vascular or biliary stricture due to mechanical and thermal injury, RFA has shown some effect on HCC with PVTT.28 Giorgio et al.29,30 introduced a technique in 2014 named “percutaneous RF thrombectomy,” which encompasses both HCC single nodules (3–5 cm) and intraportal tumoral extensions, reporting one-, three-, and five-year OS rates of 63%, 30% and 23% in 35 patients with HCC involving MPV tumor thrombus (Vp4). Conversely, the one-year survival rate in the control group (n=22 patients without any treatment) was 0% (P <0.0001), while the HCC necrosis and recanalization rate of the RFA group was 74%. Also, combined RFA and sorafenib achieved a significantly increased three-year survival rate (26%) in 49 patients as compared with that of sorafenib-alone treatment (0%) in 50 patients for HCC with Vp4 PVTT in a Western randomized control trial.17
Transarterial radioembolization (TARE) reduces the risk of hepatic parenchymal ischemia as compared to TACE by delivering iodine-131-labeled lipiodol, iodine, or yttrium-90 selectively through the hepatic arteries supplying tumor.31 Adverse effects such as fever, nausea, and abdominal pain are usually manageable by conservative support. In some retrospective studies, TARE showed better survival outcomes than sorafenib-alone treatment.32,33 Although two recent phase III randomized controlled trials did not confirm the superiority of TARE than sorafenib, several TARE treatments have been studied to increase the therapeutic effect on HCC with PVTT.34,35
Traditionally, systemic chemotherapy has not been used widely because its antitumor effect is weaker and it boasts high liver toxicity, especially in patients with liver cirrhosis. However, a recent study showed a method of infusing 5-fluorouracil (5-FU) and cisplatin into the hepatic arteries selectively in patients with advanced HCC, resulting in higher response rates and less adverse effects.36 This hepatic arterial infusion chemotherapy (HAIC) approach was relatively more effective than sorafenib-alone treatment in HCC with Vp2–4 PVTT.37
SURGICAL RESECTION OF HCC WITH PVTT
Although the recommended treatment for HCC with PVTT in BCLC classification is sorafenib, some studies have shown the better OS for surgical operation as compared with sorafenib therapy. This aggressive treatment must be individualized to the patient’s risk and tumor types.38 Depending on the location of PVTT and operator’s preference, three surgical treatments are usually adopted: hepatectomy for tumors with ipsilateral PVTT, en-bloc resection followed by portal vein reconstruction for tumors with PVTT extending beyond portal vein bifurcation, and thrombectomy for tumors with PVTT extending beyond portal vein bifurcation.39 These treatments demonstrated median OS rates of 0.91, 9.4, and 8.58 months (P =0.962), respectively, in one study with 88 patients. Another study with 113 patients with propensity score-matching reported significantly better OS in the enblock resection-treated group than in the thrombectomy–treated group (odds ratio, 1.471; P =0.017).40 Surgical resection is sometimes combined with other treatments such as adjuvant TACE, neoadjuvant RT, TARE, or concurrent chemoradiation therapy (CCRT) to prevent thrombus dissemination, lower the recurrence rate, and enhance the chance of survival.41–44 In the context of a downstaging procedure followed by surgery, the non-response of the alpha-feto protein (AFP) level seems to be a risk factor for tumor recurrence.
LIVER TRANSPLANTATION WITH OR WITHOUT DOWNSTAGING FOR HCC WITH PVTT
LT has been also not recommended due to the high recurrence rate of HCC with PVTT, similarly to surgical resection in traditional guidelines. Furthermore, LT carries the ethical consideration of applying liver grafts to the most appropriate recipients due to the shortage of liver donors. However, LT medical technology is gradually improving for patients with HCC and many centers are attempting LDLT for advanced HCC nowadays. LDLT may lower the burden of graft shortage relative to deceased donor LT.
Lee et al.45 studied 11 patients with advanced HCC who underwent LT at Seoul National University Hospital in South Korea and reported relatively good one-, three-, and five-year recurrence-free survival (RFS) rates of 63.9%, 45.5%, and 45.5% and one-, three-, and five-year OS rates of 72.7%, 63.6%, and 63.6%, respectively. Seven patients had Vp2 or 3 PVTT and four patients had Vp4 PVTT. Four patients received TACE after developing PVTT before transplantation. This study revealed some risk factors for patient death and recurrence, including MPV invasion, high AFP × ‘protein induced by the vitamin K absence/antagonist II’ (AP) score (≥20,000), high SUV ratio (tumor vs. background liver) in PET/CT (≥2.1), largest tumor size of more than 7 cm (univariate analysis due to the small case number). Among these factors, the high SUV ratio only affected the recurrence rate. Patients with low AP scores (<20,000, n=5 patients) did not experience tumor recurrence.
With increasing outcomes of various locoregional therapies on HCC with PVTT, additional studies are more focused on LT after downstaging for HCC with PVTT. A recent study of 17 patients at Asan Medical Center, South Korea with HCC and major vascular invasion received downstaging by 3D-CRT and TACE followed by LT.46 Included patients were those in which HCC had invaded the right, left portal veins or MPV; or right, middle, or left hepatic vein. Two to three weeks after TACE (2–10 mL of Lipiodol [Guerbet LLC, Princeton, NJ, USA] and 1mg/kg of cisplatin with Gelfoam [Pfizer, New York, NY, USA]), RT was performed. The planned target margin of RT was 1 to 2 cm and the total radiation dose was decided while considering liver function, residual liver volume, and adjacent organ location. Following one month of downstaging, diagnostic imaging and checking tumor markers were conducted to confirm the treatment response, and then LT was performed. Six patients received LT subsequently, while 11 patients received additional locoregional therapy, resulting in a median interval between RT and LT of 5.1 months. With a rate of 82.4% for tumor thrombus complete response, 10 among 17 patients met Milan’s criteria just before LT. One- and three-year RFS rates were 70.6% and 57.8% and one- and three-year OS rates were 87.4% and 60.5%, respectively. Notably, the three-year disease-free survival and OS rates of patients who met the Milan criteria just before LT were significantly better than those without Milan criteria (disease-free survival rate: 88.9% vs. 14.3%, P =0.007; OS rate: 80% vs. 25.7%, P =0.010). Meanwhile, another study covered five patients with HCC and Vp1–3 PVTT who received LT after downstaging involving 3D-CRT after TACE47 at Samsung Medical Center, South Korea. Here, the interval between TACE and 3D-CRT was usually two weeks (one patient did not receive TACE) and the median RFS and OS lengths were 11.7 and 35.2 months, respectively. This study also compared the outcomes between patient who received LT after downstaging and 10 patients who received RT-alone without LT by propensity score-matching, revealing significant differences in median OS times (32.5 vs. 12.2 months; P <0.01).
SBRT, which administers high-dose radiation using several beams from different angles focused on the tumor, has also been attempted for the downstaging of advanced HCC before LDLT in a recent study.48 Downstaging (SBRT with/or without TARE, TACE, or RFA) for patients with Vp3 and 4 PVTT was performed when patient’s serum bilirubin level was lower than 5 mg/dL without ascites and the nontumoral liver volume was greater than 700 mL. Study participants were re-evaluated after four to six weeks and received LDLT if there was no PVTT. For Vp1 and 2 PVTT patients, LDLT was performed 10 to 15 days after SBRT on tumor thrombus. Among 43 patients, 27 (63%) patients showed no tumor thrombus after SBRT and, finally, 25 patients received LDLT. This study compared the three groups of patients with PVTT who received LDLT after downstaging (DS group, n=25), patients with PVTT who received only LDLT without downstaging (non-DS group, n=21), and patients without PVTT who received only LDLT (upfront LT group, n=405), respectively. The DS group showed one-, three-, and five-year OS rates of 75%, 53%, and 53% and RFS rates of 78%, 78%, and 52%, respectively. Also, after censoring the two postoperative death cases which died within two months after LT, the five-year OS (57% vs. 48%) and RFS (51% vs. 40%) rates of the DS group (n=23) were slightly higher than those of the non-DS group (n=20), albeit without significant differences between them. Moreover, the five-year OS (57% vs. 65%) and RFS (51% vs. 66%) rates of the DS group were slightly lower than those of the upfront LT group after censoring postoperative death, again without significant differences. In the DS group, tumor grade III/IV was a risk factor for reduced OS, while high preoperative AFP (>400 ng/mL) and low delta AFP (AFP change between before and after downstaging, <2,000 ng/mL) were poor prognostic factors for RFS. Meanwhile, there are other studies of downstaging prior to LT showing acceptable outcomes;49,50 one study noted that the median RFS time of four patients who received TARE as downstaging was 39 months.51
CCRT was also attempted as a downstaging method of HCC with PVTT before LDLT.52 In one study, RT combined with 5-FU was administered during five weeks and additional HAIC (5-FU/cisplatin combination) was introduced for about three to 12 months in four-week intervals at Severance Hospital, South Korea. All eight patients achieved successful downstaging, with no PVTT, and received LDLT. The one-year OS rate was 87.5% and the median survival time was 33 months.
Immunotherapy is not yet widely used for downstaging method of HCC with PVTT before LT. However, there is a case report of using nivolumab, an immune checkpoint inhibitor before LT in a patient with HCC outside Milan criteria (due to 4 lesions in liver) resulting patient surviving more than one-year without tumor recurrence.53 This result showed the potential for the role of immunotherapy in downstaging method of HCC with PVTT. In this setting, immune therapy just prior to or after LT may be fatal due to 4-week half-life and hepatotoxicity of nivolumab.54
CONCLUSION
To this day, the American Association for the Study of Liver Disease and European Association for the Study of the Liver guidelines do not offer suggestions for LT after downstaging for HCC with PVTT.55,56 However, given that more recent studies of downstaging procedures have promoted favorable outcomes, LT after downstaging is increasingly worth considering for HCC with PVTT. Although these trials are not of excellent quality, this approach still shows better outcomes than sorafenib-alone treatment. Due to the small number of existing studies, more trials and evidence are needed before stronger conclusions can be drawn.
There are a few common things that should be considered. First, the optimal downstaging protocol is important to the outcome. RT, TACE, or TARE can be considered according to patients and tumor status. Second, risk factors affecting OS and RFS rates should be considered. The predownstaging AFP value, delta AFP value, disappearance of PVTT on imaging, and meeting the Milan criteria or not after downstaging may be considered.
Therefore, for HCC with PVTT, locoregional therapy with LT in mind would be helpful. After that, if the reaction of the downstaged tumor is good, then LT may be performed in selected cases; otherwise, other therapies can be considered.
Notes
Conflicts of Interest
The authors declare no conflict of interest.
Ethics Statement
This review article is fully based on the articles which was already published and did not involve additional patient participants. Therefore, IRB approval is not necessary.
Funding Statement
No funding to declare.
Data Availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the study.
Author Contribution
Conception and design, data acquisition, drafting the article: SJK.
Conception and design, critical revision of the article: JMK.
All authors have reviewed and approved the final version of manuscript.
