Articles
- Page Path
- HOME > J Liver Cancer > Volume 20(1); 2020 > Article
-
Review Article
Histopathological Variants of Hepatocellular Carcinomas: an Update According to the 5th Edition of the WHO Classification of Digestive System Tumors -
Haeryoung Kim1, Mi Jang2, Young Nyun Park2

-
Journal of Liver Cancer 2020;20(1):17-24.
DOI: https://doi.org/10.17998/jlc.20.1.17
Published online: March 31, 2020
1Department of Pathology, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea
2Department of Pathology, Brain Korea 21 PLUS Project for Medical Science, Yonsei University College of Medicine, Seoul, Korea
-
Corresponding author : Young Nyun Park Department of Pathology, Yonsei University College of Medicine, 50 Yonsei-ro, Seodaemun-gu, Seoul 03722, Korea
Tel. +82-2-2228-1678, Fax. +82-2-362-0860 E-mail; young0608@yuhs.ac
Copyright © 2020 The Korean Liver Cancer Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 14,104 Views
- 1,000 Downloads
- 26 Citations
Abstract
- Hepatocellular carcinoma (HCC) is heterogeneous in pathogenesis, phenotype and biological behavior. Various histopathological features of HCC had been sporadically described, and with the identification of common molecular alterations of HCC and its genomic landscape over the last decade, morpho-molecular correlation of HCC has become possible. As a result, up to 35% of HCCs can now be classified into histopathological variants, many of which have unique molecular characteristics. This review will provide an introduction to the variously described histopathological variants of HCC in the updated WHO Classification of Digestive System Tumors.
- Hepatocellular carcinoma (HCC) is defined as a primary hepatic malignancy showing hepatocellular differentiation, and it accounts for 75-85% of primary liver cancers [1]. HCCs mostly arise in a background of chronic liver disease, the most common etiologies being hepatitis B, hepatitis C, chronic alcohol abuse, non-alcoholic fatty liver disease, inherited diseases (e.g. hemochromatosis and glycogen storage disease), and exogenous substances, such as aflatoxin B1 [1,2]. A minority of HCCs develop in a background of normal or near-normal liver, the most common setting being HCCs arising in hepatocellular adenomas [1,3,4].
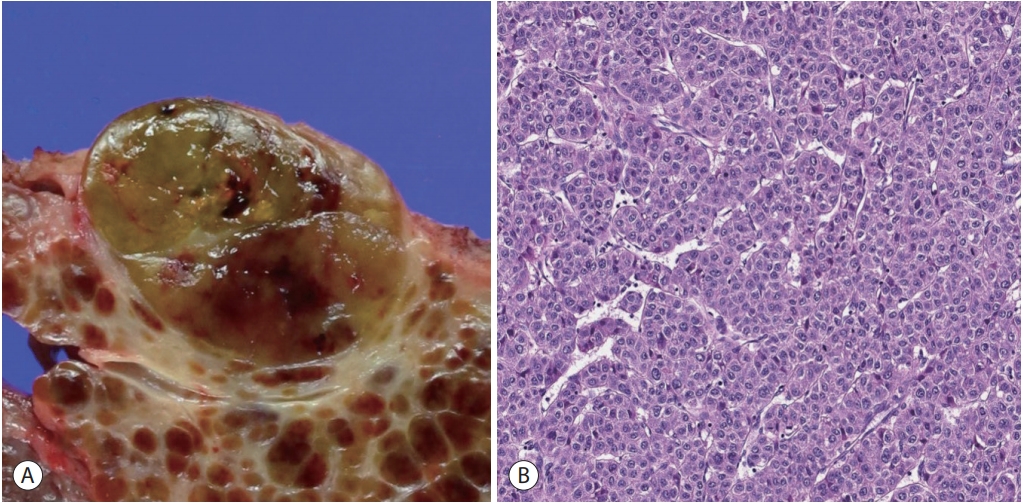
- Histologically, conventional HCCs demonstrate hepatocytic differentiation (i.e., the tumor cells resemble the appearance of hepatocytes with varying degrees of cyto-architectural atypia). The typical HCC tumor cells are cuboidal in shape, contain abundant eosinophilic cytoplasm with centrally located nuclei, and are frequently arranged in a trabecular pattern of variable thickness that at least vaguely recapitulates the trabecular architecture of the normal hepatic acinus, with very little intratumoral stroma (Fig. 1) [5]. However, many cases demonstrate deviations from this pattern; for example, some HCCs are predominantly composed of tumor cells with clear cytoplasm, some may show a predominantly pseudoglandular architecture and some may show abundant intratumoral fibrous stroma.
- The 4th Edition of the World Health Organization (WHO) Classification of Digestive System Tumors (2010) described the histopathological features of HCC in detail, including detailed descriptions of “cytological variants”, such as pleomorphic cells, clear cells and spindle cells [6]. In addition, a few “special types” of HCC were recognized, including fibrolamellar carcinoma, scirrhous HCC, undifferentiated carcinoma, lymphoepithelioma-like carcinoma and sarcomatoid HCC. At that time, these special types were based mostly on the morphological features of the tumor.
- Since then, numerous large-scale genomic analyses in the recent years have clarified the mutational landscape and identified key cell-signaling and metabolic pathways related to hepatocarcinogenesis [7-10]. This has resulted in a large number of molecular subclasses and signatures of HCC, and has highlighted the heterogeneity of HCC, in pathogenesis, phenotype and biological behavior. Recent studies have provided increasing evidence for morpho-molecular correlation of HCCs, and these efforts have resulted in several proposed histopathological variants of HCC [11]. As many as 35% of HCCs can now be classified into histopathological variants, which have been introduced in the latest WHO Classification of Digestive System Tumors, 5th Edition [1] and the distinction between most of these variants is now supported by the molecular features of the tumors (Table 1). In this review, we will summarize the recently described histopathological variants of HCCs.
INTRODUCTION
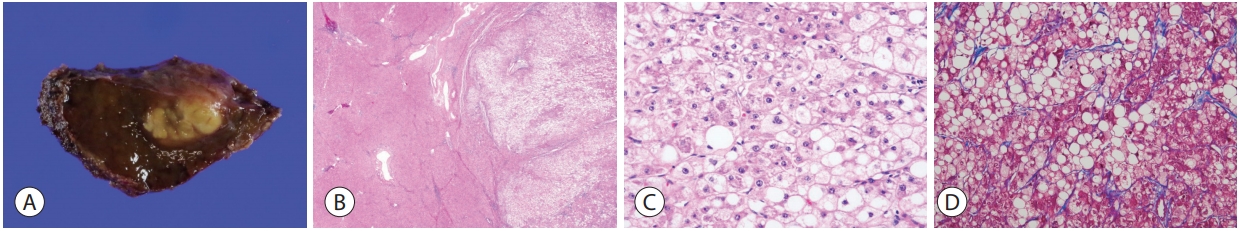
- The steatohepatitic variant of HCC, or steatohepatitic HCC, demonstrates the histological features of steatohepatitis within the tumor, including steatosis, ballooning of tumor cells, inflammation and the typical “chicken-wire pattern” pericellular fibrosis (Fig. 2) [11-14]. It accounts for 5-20% of HCCs, and the background liver may show steatohepatitis [13,14]. Although this variant has been shown to be less often associated with vascular invasion or satellite nodules, its prognosis seems to be similar to conventional HCCs so far [11]. The key molecular features include IL-6/JAK/STAT activation, and lower frequency of CTNNB1 , TERT , and TP53 mutations compared to other HCCs [11].
STEATOHEPATITIC VARIANT
- In the clear cell variant of HCC, the majority (>80%) of tumor cells demonstrate clear cytoplasm (Fig. 3) [15]. The clear cytoplasm is a result of glycogen accumulation, although it is sometimes hard to discriminate from lipid droplets that are intermixed; in this regard, some degree of steatosis is acceptable for a diagnosis of clear cell HCC. About 3-7% of HCCs show this morphology, and although the prognosis has been shown to be better compared to conventional HCCs, there is still a lack of information on the clinical correlates or the molecular features of these tumors. Of note, it is important to discriminate clear cell HCC from metastatic clear cell renal cell carcinoma, as the morphology may be remarkably similar. Therefore, on encountering such cases in a biopsy, it is advisable to check the patient’s clinicoradiological information, and to consider performing immunohistochemical stains (e.g., PAX-8, CD10, RCC for renal cell carcinoma, and hepatocellular differentiation markers such as HepPar-1 and arginase-1 for HCC) to avoid this diagnostic pitfall [16].
CLEAR CELL VARIANT
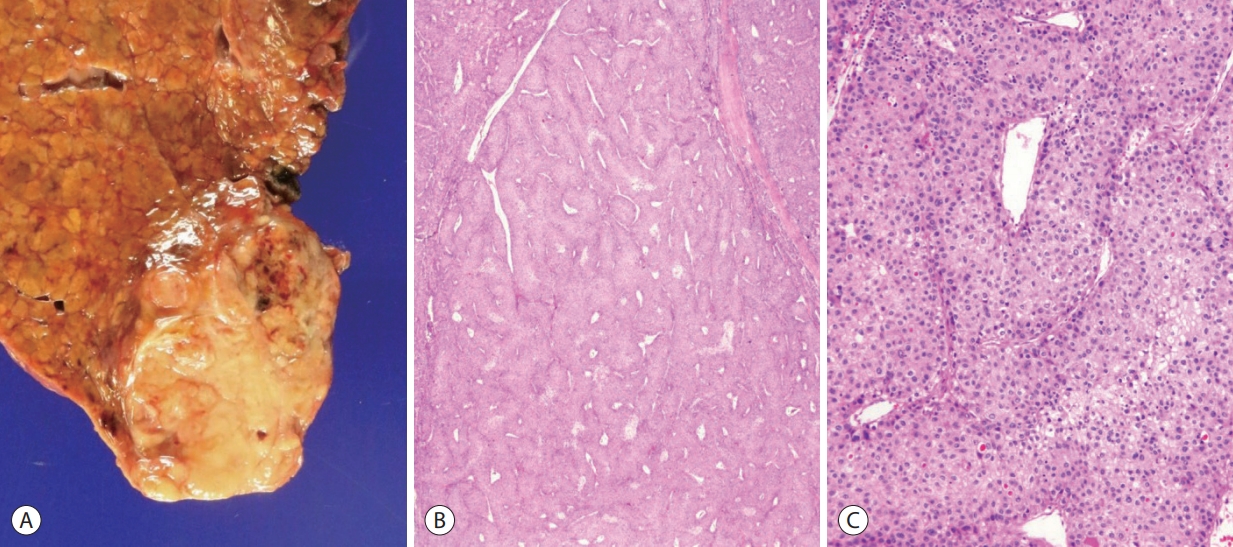
- The macrotrabecular-massive variant of HCC demonstrates a characteristic microscopic appearance: prominent thick trabeculae, measuring more than 6-10 cells in thickness, in at least 50% of the tumor (Fig. 4) [1,11,17]. The relative frequency of this variant has been described to be about 5% in the WHO classification, although some have reported frequencies of up to 20%, probably as a result of the varying histological criteria; the trabecular thickness criteria for macrotrabecular- massive HCC has varied from 6 to 20 cells thick in the literature. From the molecular level, this variant is characterized by frequent TP53 mutation and FGF19 amplification [11,17]. Macrotrabecular-massive HCCs have also been associated with frequent HBV infection, frequent vascular invasion, poor differentiation, CK19 expression, high serum AFP levels and a poor clinical outcome [11,17].
MACROTRABECULAR-MASSIVE VARIANT
- Scirrhous HCC is characterized by dense intratumoral fibrous stroma, occupying at least 30-50% of the tumor (Fig. 5) [11,18-20]. The tumor cell nests are predominantly composed of mature hepatocyte-like cells and rimmed by smaller tumor cells that resemble hepatic stem/progenitor cells – the latter population frequently express stemness-related markers, such as CK19, CK7, and epithelial cell adhesion molecule [19,20]. This variant accounts for 4% of HCCs, and the dense fibrous stroma results in imaging findings that overlap with those of cholangiocarcinoma. TSC1/TSC2 mutations and transforming growth factor-beta signaling activation have been demonstrated in these tumors [11,20]. The clinical outcome of this variant is still controversial. However, scirrhous HCCs have been reported to demonstrate decreased survival compared to conventional HCCs, especially in larger tumors (≥5 cm), and aggressive pathological features (e.g. vascular invasion, infiltrative growth) have been more frequently seen in these tumors compared to conventional HCCs [20,21].
SCIRRHOUS VARIANT
- This is a rare (up to 3%) variant of HCC that is characterized by clear (chromophobic) cytoplasm of the tumor cells, and focal areas of striking nuclear atypia in a background of otherwise “bland-looking” cytology, and thus have been initially named “chromophobe HCC with abrupt anaplasia” [22]. From a molecular viewpoint, it is interesting that the majority of the reported cases demonstrated alternative lengthening of telomere phenotype by telomere fluorescence in situ hybridization (FISH) [22]. Little is known regarding the clinical correlates of this variant, and the prognosis so far seems to be similar to conventional HCCs.
CHROMOPHOBE VARIANT
- The fibrolamellar subtype of HCC had already been designated as a subtype of HCC in the previous WHO classification [6], and has unique clinical characteristics, including the age distribution (children and young adults) and the absence of underlying chronic liver disease [1,23]. It is a rare subtype that accounts for up to 1% of HCCs and occurs more frequently in Western countries [1]. Histologically, it is characterized by parallel arrays of large eosinophilic tumour cells with large vesicular nuclei and prominent nucleoli, and separated by dense fibrous septa. Due to the abundance of fibrous intratumoral stroma, it may appear similar to scirrhous HCC; however, the fibrous stroma of scirrhous HCC has been recently shown to contain more abundant cancer-associated fibroblasts and tumor-associated macrophages, and stemness-related marker expression is more frequently seen in scirrhous HCCs.24 Recently, DNAJB1-PRKACA gene fusion has been demonstrated in fibrolamellar HCC, and hence FISH for PRKACA gene rearrangement has been proposed to be a good diagnostic tool for this tumor [25,26]. The prognosis of fibrolamellar HCC has been shown to be better than for conventional HCCs that arise in cirrhotic livers, but similar to HCCs in non-cirrhotic livers.27 In addition, conventional HCCs with features of fibrolamellar HCC in parts of the tumor have been shown to have similar prognosis with conventional HCCs, suggesting that a strict criterion (fibrolamellar features in the entire tumor) is required for a diagnosis of fibrolamellar HCC [28].
FIBROLAMELLAR HCC (FIBROLAMELLAR CARCINOMA)
- This is a very rare variant of HCCs (less than 1%) that is characterized histologically by diffuse neutrophilic infiltration within the HCC, and clinically by elevated leukocyte counts, C-reactive protein and IL-6, and a poor prognosis compared to conventional HCCs. The tumor cells may have focal sarcomatoid appearance, and granulocyte colony-stimulating factor production by the tumor cells have been reported [29].
NEUTROPHIL-RICH VARIANT
- The lymphocyte-rich variant of HCC has been shown to account for <1% of HCCs, and demonstrates massive intratumoral lymphocytic infiltration (Fig. 6). It has also been referred to as lymphoepithelioma-like HCCs [29,30]. These tumors have been associated with a favorable clinical outcome and also more frequent programmed death-ligand 1 expression [31-33]. The molecular features of this subtype have not been characterized yet; however, an association with Epstein-Barr virus infection has not been demonstrated, unlike similar tumors of some other organs such as the stomach [32]. Interestingly, a minority of HCCs also demonstrates lymphoid aggregates or follicles within the tumor, and the presence of these tertiary lymphoid structures has been associated with a favorable prognosis [34].
LYMPHOCYTE-RICH VARIANT
- Following the accumulation of clinicopathological studies on various histopathological forms of HCC and the increasing number of molecular studies addressing the heterogeneity of HCC, there have been efforts to connect the histomorphological and molecular features of HCCs, and such links have resulted in the currently proposed HCC variants of the recent WHO Classification of Digestive System Tumors [1]. With the recent morpho-molecular correlations of HCCs and the identification of various histopathological variants of HCC, most of which are based on their molecular features, we can now not only better understand the pathogenesis of HCC, but also use this knowledge to predict patient outcome and to facilitate the development of targeted therapy. More importantly, the identification of histopathological variants of HCCs suggests that histopathology still has an important role in HCC patient management.
CONCLUSION
Acknowledgments



WHO, World Health Organization; IL, interleukin; JAK, janus kinase; STAT, signal transducer and activator of transcription; NAFLD, non-alcoholic fatty liver disease; HCC, hepatocellular carcinoma; AFP, alpha-fetoprotein; TGF-β, transforming growth factor-beta; ALT, alternative lengthening of telomere; G-CSF, granulocyte colony stimulating factor; PD-L1, programmed death-ligand 1; EBV, Epstein-Barr virus.
- 1. Torbenson MS, Ng IOL, Park YN, Roncalli M, Sakamoto M. Hepatocellular carcinoma. In: WHO Classification of Tumours Editorial Board, editor. Digestive system tumours. WHO classification of tumours series. 5th ed. Lyon: International Agency for Research on Cancer; 2019;229−39.Article
- 2. Sanyal AJ, Yoon SK, Lencioni R. The etiology of hepatocellular carcinoma and consequences for treatment. Oncologist 2010;15 Suppl 4:14−22.ArticlePubMed
- 3. Degasperi E, Colombo M. Distinctive features of hepatocellular carcinoma in non-alcoholic fatty liver disease. Lancet Gastroenterol Hepatol 2016;1:156−164.ArticlePubMed
- 4. Nault JC, Mallet M, Pilati C, Calderaro J, Bioulac-Sage P, Laurent C, et al. High frequency of telomerase reverse-transcriptase promoter somatic mutations in hepatocellular carcinoma and preneoplastic lesions. Nat Commun 2013;4:2218. ArticlePubMedPMCPDF
- 5. Ferrell LD, Kakar S, Terracciano LM, Wee A. Tumours and tumourlike lesions of the liver. Edited by Burt AD, Ferrell LD, Hubscher SG: MacSween’s Pathology of the Liver. 7th ed. Philadelphia, Philadelphia: Elsevier. 2018, pp 780−879.
- 6. Theise ND, Curado MP, Franceschi S, Hytiroglou P, Kudo M, Park YN, et al. Hepatocellular Carcinoma. Edited by Bosman FT, Carneiro F, Hruban RH, Theise RH: WHO classification of tumours of the digestive system. 4th ed. Lyon, IARC. 2010, pp 205−16.
- 7. Hoshida Y, Nijman SM, Kobayashi M, Chan JA, Brunet JP, Chiang DY, et al. Integrative transcriptome analysis reveals common molecular subclasses of human hepatocellular carcinoma. Cancer Res 2009;69:7385−7392.ArticlePubMedPMC
- 8. Boyault S, Rickman DS, de Reyniès A, Balabaud C, Rebouissou S, Jeannot E, et al. Transcriptome classification of HCC is related to gene alterations and to new therapeutic targets. Hepatology 2007;45:42−52.ArticlePubMed
- 9. Chiang DY, Villanueva A, Hoshida Y, Peix J, Newell P, Minguez B, et al. Focal gains of VEGFA and molecular classification of hepatocellular carcinoma. Cancer Res 2008;68:6779−6788.ArticlePubMedPMC
- 10. Lee JS, Chu IS, Heo J, Calvisi DF, Sun Z, Roskams T, et al. Classification and prediction of survival in hepatocellular carcinoma by gene expression profiling. Hepatology 2004;40:667−676.ArticlePubMed
- 11. Calderaro J, Couchy G, Imbeaud S, Amaddeo G, Letouzé E, Blanc JF, et al. Histological subtypes of hepatocellular carcinoma are related to gene mutations and molecular tumour classification. J Hepatol 2017;67:727−738.ArticlePubMed
- 12. Lee JS, Yoo JE, Kim H, Rhee H, Koh MJ, Nahm JH, et al. Tumor stroma with senescence-associated secretory phenotype in steatohepatitic hepatocellular carcinoma. PLoS One 2017;12:e0171922.ArticlePubMedPMC
- 13. Salomao M, Remotti H, Vaughan R, Siegel AB, Lefkowitch JH, Moreira RK. The steatohepatitic variant of hepatocellular carcinoma and its association with underlying steatohepatitis. Hum Pathol 2012;43:737−746.ArticlePubMed
- 14. Salomao M, Yu WM, Brown RS Jr, Emond JC, Lefkowitch JH. Steatohepatitic hepatocellular carcinoma (SH-HCC): a distinctive histological variant of HCC in hepatitis C virus-related cirrhosis with associated NAFLD/NASH. Am J Surg Pathol 2010;34:1630−1636.ArticlePubMed
- 15. Li T, Fan J, Qin LX, Zhou J, Sun HC, Qiu SJ, et al. Risk factors, prognosis, and management of early and late intrahepatic recurrence after resection of primary clear cell carcinoma of the liver. Ann Surg Oncol 2011;18:1955−1963.ArticlePubMedPDF
- 16. Reuter VE, Argani P, Zhou M, Delahunt B; Members of the ISUP Immunohistochemistry in Diagnostic Urologic Pathology Group. Best practices recommendations in the application of immunohistochemistry in the kidney tumors: report from the International Society of Urologic Pathology consensus conference. Am J Surg Pathol 2014;38:e35−e49.ArticlePubMed
- 17. Ziol M, Poté N, Amaddeo G, Laurent A, Nault JC, Oberti F, et al. Macrotrabecular-massive hepatocellular carcinoma: a distinctive histological subtype with clinical relevance. Hepatology 2018;68:103−112.ArticlePubMed
- 18. Kurogi M, Nakashima O, Miyaaki H, Fujimoto M, Kojiro M. Clinicopathological study of scirrhous hepatocellular carcinoma. J Gastroenterol Hepatol 2006;21:1470−1477.ArticlePubMed
- 19. Matsuura S, Aishima S, Taguchi K, Asayama Y, Terashi T, Honda H, et al. ‘Scirrhous’ type hepatocellular carcinomas: a special reference to expression of cytokeratin 7 and hepatocyte paraffin 1. Histopathology 2005;47:382−390.ArticlePubMed
- 20. Seok JY, Na DC, Woo HG, Roncalli M, Kwon SM, Yoo JE, et al. A fibrous stromal component in hepatocellular carcinoma reveals a cholangiocarcinoma-like gene expression trait and epithelialmesenchymal transition. Hepatology 2012;55:1776−1786.ArticlePubMed
- 21. Kim SH, Lim HK, Lee WJ, Choi D, Park CK. Scirrhous hepatocellular carcinoma: comparison with usual hepatocellular carcinoma based on CT-pathologic features and long-term results after curative resection. Eur J Radiol 2009;69:123−130.ArticlePubMed
- 22. Wood LD, Heaphy CM, Daniel HD, Naini BV, Lassman CR, Arroyo MR, et al. Chromophobe hepatocellular carcinoma with abrupt anaplasia: a proposal for a new subtype of hepatocellular carcinoma with unique morphological and molecular features. Mod Pathol 2013;26:1586−1593.ArticlePubMedPMCPDF
- 23. Chagas AL, Kikuchi L, Herman P, Alencar RS, Tani CM, Diniz MA, et al. Clinical and pathological evaluation of fibrolamellar hepatocellular carcinoma: a single center study of 21 cases. Clinics (Sao Paulo) 2015;70:207−213.ArticlePubMedPMCPDF
- 24. Kim YJ, Rhee H, Yoo JE, Alves VAF, Kim GJ, Kim HM, et al. Tumour epithelial and stromal characteristics of hepatocellular carcinomas with abundant fibrous stroma: fibrolamellar versus scirrhous hepatocellular carcinoma. Histopathology 2017;71:217−226.ArticlePubMed
- 25. Graham RP, Jin L, Knutson DL, Kloft-Nelson SM, Greipp PT, Waldburger N, et al. DNAJB1-PRKACA is specific for fibrolamellar carcinoma. Mod Pathol 2015;28:822−829.ArticlePubMedPDF
- 26. Honeyman JN, Simon EP, Robine N, Chiaroni-Clarke R, Darcy DG, Lim II, et al. Detection of a recurrent DNAJB1-PRKACA chimeric transcript in fibrolamellar hepatocellular carcinoma. Science 2014;343:1010−1014.ArticlePubMedPMC
- 27. Kakar S, Burgart LJ, Batts KP, Garcia J, Jain D, Ferrell LD. Clinicopathologic features and survival in fibrolamellar carcinoma: comparison with conventional hepatocellular carcinoma with and without cirrhosis. Mod Pathol 2005;18:1417−1423.ArticlePubMedPDF
- 28. Malouf GG, Brugières L, Le Deley MC, Faivre S, Fabre M, Paradis V, et al. Pure and mixed fibrolamellar hepatocellular carcinomas differ in natural history and prognosis after complete surgical resection. Cancer 2012;118:4981−4990.ArticlePubMed
- 29. Torbenson MS. Morphologic subtypes of hepatocellular carcinoma. Gastroenterol Clin North Am 2017;46:365−391.ArticlePubMed
- 30. Calderaro J, Ziol M, Paradis V, Zucman-Rossi J. Molecular and histological correlations in liver cancer. J Hepatol 2019;71:616−630.ArticlePubMed
- 31. Sia D, Jiao Y, Martinez-Quetglas I, Kuchuk O, Villacorta-Martin C, Castro de Moura M, et al. Identification of an immune-specific class of hepatocellular carcinoma, based on molecular features. Gastroenterology 2017;153:812−826.ArticlePubMed
- 32. Chan AW, Tong JH, Pan Y, Chan SL, Wong GL, Wong VW, et al. Lymphoepithelioma-like hepatocellular carcinoma: an uncommon variant of hepatocellular carcinoma with favorable outcome. Am J Surg Pathol 2015;39:304−312.ArticlePubMed
- 33. Calderaro J, Rousseau B, Amaddeo G, Mercey M, Charpy C, Costentin C, et al. Programmed death ligand 1 expression in hepatocellular carcinoma: relationship with clinical and pathological features. Hepatology 2016;64:2038−2046.ArticlePubMed
- 34. Calderaro J, Petitprez F, Becht E, Laurent A, Hirsch TZ, Rousseau B, et al. Intra-tumoral tertiary lymphoid structures are associated with a low risk of early recurrence of hepatocellular carcinoma. J Hepatol 2019;70:58−65.ArticlePubMed
References
Figure & Data
References
Citations

- Computational pathology: A survey review and the way forward
Mahdi S. Hosseini, Babak Ehteshami Bejnordi, Vincent Quoc-Huy Trinh, Lyndon Chan, Danial Hasan, Xingwen Li, Stephen Yang, Taehyo Kim, Haochen Zhang, Theodore Wu, Kajanan Chinniah, Sina Maghsoudlou, Ryan Zhang, Jiadai Zhu, Samir Khaki, Andrei Buin, Fatemeh
Journal of Pathology Informatics.2024; 15: 100357. CrossRef - Characterization of lymphocyte‐rich hepatocellular carcinoma and the prognostic role of tertiary lymphoid structures
Bokyung Ahn, Hee‐Sung Ahn, Jinho Shin, Eunsung Jun, Eun‐Young Koh, Yeon‐Mi Ryu, Sang‐Yeob Kim, Chang Ohk Sung, Ju Hyun Shim, JeongYeon Hong, Kyunggon Kim, Hyo Jeong Kang
Liver International.2024; 44(5): 1202. CrossRef - Clinical‐Radiologic Morphology‐Radiomics Model on Gadobenate Dimeglumine‐Enhanced MRI for Identification of Highly Aggressive Hepatocellular Carcinoma: Temporal Validation and Multiscanner Validation
Wanjing Zheng, Xiaodan Chen, Meilian Xiong, Yu Zhang, Yang Song, Dairong Cao
Journal of Magnetic Resonance Imaging.2024;[Epub] CrossRef - Low-Baseline PD1+ Granulocytes Predict Responses to Atezolizumab–Bevacizumab in Hepatocellular Carcinoma
Catia Giovannini, Fabrizia Suzzi, Francesco Tovoli, Mariangela Bruccoleri, Mariarosaria Marseglia, Eleonora Alimenti, Francesca Fornari, Massimo Iavarone, Fabio Piscaglia, Laura Gramantieri
Cancers.2023; 15(6): 1661. CrossRef - Non-alcoholic fatty liver disease: the pathologist’s perspective
Wei-Qiang Leow, Anthony Wing-Hung Chan, Paulo Giovanni L. Mendoza, Regina Lo, Kihan Yap, Haeryoung Kim
Clinical and Molecular Hepatology.2023; 29(Suppl): S302. CrossRef - 2022 KLCA-NCC Korea practice guidelines for the management of hepatocellular carcinoma
Journal of Liver Cancer.2023; 23(1): 1. CrossRef - Three-dimensional multifrequency magnetic resonance elastography improves preoperative assessment of proliferative hepatocellular carcinoma
Guixue Liu, Di Ma, Huafeng Wang, Jiahao Zhou, Zhehan Shen, Yuchen Yang, Yongjun Chen, Ingolf Sack, Jing Guo, Ruokun Li, Fuhua Yan
Insights into Imaging.2023;[Epub] CrossRef - Lymphocyte-Rich Hepatocellular Carcinoma with Multiple Lymphadenopathy and Positive Epstein–Barr Virus Encoding Region
Pin-Yi Wang, Yu-Hsuan Kuo, Ming-Jen Sheu, Hsing-Tao Kuo, Wen-Ying Lee, Yu-Ting Kuo, Su-Hung Wang, Zu-Yau Lin
Case Reports in Hepatology.2023; 2023: 1. CrossRef - Genome-Wide Extrachromosomal Circular DNA Profiling of Paired Hepatocellular Carcinoma and Adjacent Liver Tissues
Jianyu Ye, Peixin Huang, Kewei Ma, Zixin Zhao, Ting Hua, Wenjing Zai, Jieliang Chen, Xiutao Fu
Cancers.2023; 15(22): 5309. CrossRef - Proliferative hepatocellular carcinomas in cirrhosis: patient outcomes of LI-RADS category 4/5 and category M
Subin Heo, Hyo Jeong Kang, Sang Hyun Choi, Sehee Kim, Youngeun Yoo, Won-Mook Choi, So Yeon Kim, Seung Soo Lee
European Radiology.2023;[Epub] CrossRef - Cellular heterogeneity and plasticity in liver cancer
Lo-Kong Chan, Yu-Man Tsui, Daniel Wai-Hung Ho, Irene Oi-Lin Ng
Seminars in Cancer Biology.2022; 82: 134. CrossRef - MRI features of histologic subtypes of hepatocellular carcinoma: correlation with histologic, genetic, and molecular biologic classification
Ja Kyung Yoon, Jin-Young Choi, Hyungjin Rhee, Young Nyun Park
European Radiology.2022; 32(8): 5119. CrossRef - Variant Hepatocellular Carcinoma Subtypes According to the 2019 WHO Classification: An Imaging-Focused Review
Liang Meng Loy, Hsien Min Low, Jin-Young Choi, Hyungjin Rhee, Chin Fong Wong, Cher Heng Tan
American Journal of Roentgenology.2022; 219(2): 212. CrossRef - Paraneoplastic syndromes in hepatocellular carcinoma: a review
Yuki Ong, Cheong Wei Terence Huey, Vishalkumar Girishchandra Shelat
Expert Review of Gastroenterology & Hepatology.2022; 16(5): 449. CrossRef - Morphomolecular Classification Update on Hepatocellular Adenoma, Hepatocellular Carcinoma, and Intrahepatic Cholangiocarcinoma
Venkata S. Katabathina, Lokesh Khanna, Venkateswar R. Surabhi, Marta Minervini, Krishna Shanbhogue, Anil K. Dasyam, Srinivasa R. Prasad
RadioGraphics.2022; 42(5): 1338. CrossRef - 2022 KLCA-NCC Korea practice guidelines for the management of hepatocellular carcinoma
Clinical and Molecular Hepatology.2022; 28(4): 583. CrossRef - 2022 KLCA-NCC Korea Practice Guidelines for the Management of Hepatocellular Carcinoma
Korean Journal of Radiology.2022; 23(12): 1126. CrossRef - Histomorphological Subtypes of Hepatocellular Carcinoma and Intrahepatic Cholangiocarcinoma: Review and Update
Yoon Jung Hwang, Haeryoung Kim
AJSP: Reviews and Reports.2022; 27(6): 234. CrossRef - Pediatric Primary Hepatic Tumors: Diagnostic Considerations
Bryony Lucas, Sanjita Ravishankar, Irina Pateva
Diagnostics.2021; 11(2): 333. CrossRef - A Case of Lymphocyte-Rich Hepatocellular Carcinoma in a Patient Who Was Treated for Colon Cancer
Jae Won Song, Ho Soo Chun, Jae Seung Lee, Hye Won Lee, Beom Kyung Kim, Seung Up Kim, Jun Yong Park, Sang Hoon Ahn, Young Nyun Park, Dai Hoon Han, Do Young Kim
Journal of Liver Cancer.2021; 21(1): 69. CrossRef - HCC You Cannot See
Vaishnavi Boppana, Sakshi Sahni, Joseph Glass, Christopher Chang, Denis M McCarthy
Digestive Diseases and Sciences.2021; 66(7): 2185. CrossRef - An update on subtypes of hepatocellular carcinoma: From morphology to molecular
Monika Vyas, Dhanpat Jain
Indian Journal of Pathology and Microbiology.2021; 64(5): 112. CrossRef - HCC: role of pre- and post-treatment tumor biology in driving adverse outcomes and rare responses to therapy
Sandeep Arora, Roberta Catania, Amir Borhani, Natally Horvat, Kathryn Fowler, Carla Harmath
Abdominal Radiology.2021; 46(8): 3686. CrossRef - Gadoxetate-enhanced MRI Features of Proliferative Hepatocellular Carcinoma Are Prognostic after Surgery
Hyo-Jin Kang, Haeryoung Kim, Dong Ho Lee, Bo Yun Hur, Yoon Jung Hwang, Kyung-Suk Suh, Joon Koo Han
Radiology.2021; 300(3): 572. CrossRef - Radiologic Diagnosis of Hepatocellular Carcinoma
Woo Kyoung Jeong
The Korean Journal of Gastroenterology.2021; 78(5): 261. CrossRef - Update on Hepatocellular Carcinoma: a Brief Review from Pathologist Standpoint
Nese Karadag Soylu
Journal of Gastrointestinal Cancer.2020; 51(4): 1176. CrossRef

 E-submission
E-submission THE KOREAN LIVER CANCER ASSOCIATION
THE KOREAN LIVER CANCER ASSOCIATION
 PubReader
PubReader ePub Link
ePub Link Download Citation
Download Citation

 Follow JLC on Twitter
Follow JLC on Twitter