Search
- Page Path
- HOME > Search
Original Article
- The dual role of transforming growth factor-beta signatures in human B viral multistep hepatocarcinogenesis: early and late responsive genes
- Jeong Eun Yoo, Ji Hae Nahm, Young-Joo Kim, Youngsic Jeon, Young Nyun Park
- J Liver Cancer. 2022;22(2):115-124. Published online May 20, 2022
- DOI: https://doi.org/10.17998/jlc.2022.04.20
- 2,426 Views
- 99 Downloads
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background/Aim
Transforming growth factor-beta (TGF-β) has a dichotomous role, functioning as a tumor suppressor and tumor promoter. TGF-β signatures, explored in mouse hepatocytes, have been reported to predict the clinical outcomes of hepatocellular carcinoma (HCC) patients; HCCs exhibiting early TGF-β signatures showed a better prognosis than those with late TGF-β signatures. The expression status of early and late TGF-β signatures remains unclear in defined lesions of human B-viral multistep hepatocarcinogenesis.
Methods
The expression of TGF-β signatures, early and late responsive signatures of TGF-β were investigated and analyzed for their correlation in cirrhosis, low-grade dysplastic nodules (DNs), high-grade DNs, early HCCs and progressed HCCs (pHCCs) by real-time PCR and immunohistochemistry.
Results
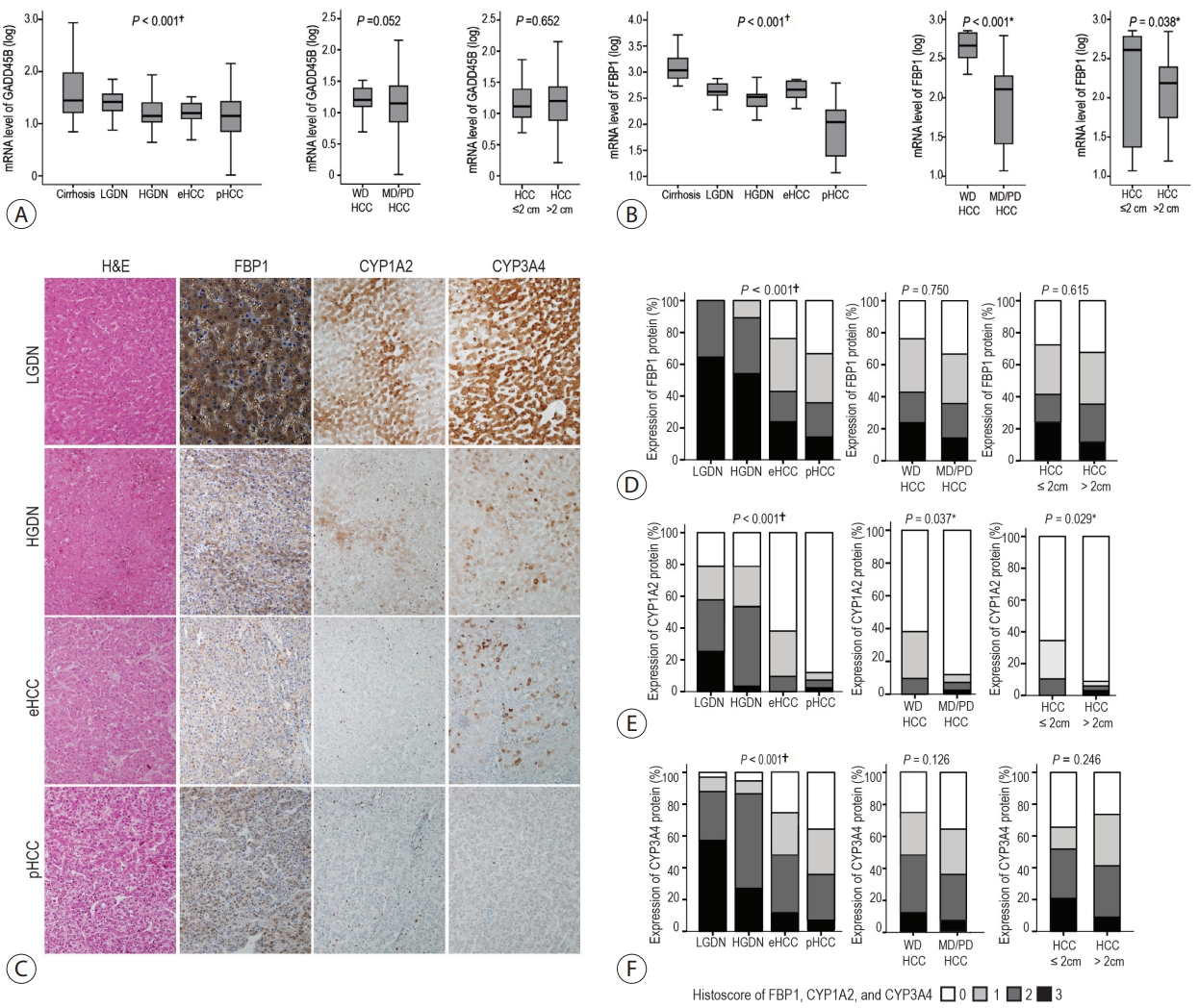
The expression levels of TGF-β signaling genes (TGFB1, TGFBR1, TGFBR2 and SMAD4) gradually increased with the progression of hepatocarcinogenesis, peaking in pHCCs. The expression of early responsive genes of TGF-β (GADD45B, FBP1, CYP1A2 and CYP3A4) gradually decreased, and that of the late TGF-β signatures (TWIST and SNAI1) significantly increased according to the progression of multistep hepatocarcinogenesis. Furthermore, mRNA levels of TWIST and SNAI1 were well correlated with those of stemness markers, with upregulation of TGF-β signaling, whereas FBP1 expression was inversely correlated with that of stemness markers.
Conclusions
The enrichment of the late responsive signatures of TGF-β with induction of stemness is considered to be involved in the progression of the late stage of multistep hepatocarcinogenesis, whereas the early responsive signatures of TGF-β are suggested to have tumor-suppressive roles in precancerous lesions of the early stage of multistep hepatocarcinogenesis.

Review Article
- Genomic Heterogeneity of Hepatocellular Carcinomas
- Hyun Goo Woo
- Journal of the Korean Liver Cancer Study Group. 2013;13(1):48-50. Published online February 28, 2013
- DOI: https://doi.org/10.17998/jlc.13.1.48
- 874 Views
- 5 Downloads
-
 Abstract
Abstract
 PDF
PDF - Hepatocellular carcinoma (HCC) and cholangiocarcinoma (CC) are major primary liver cancers in adults, comprising liver cancer spectrum. The existence of combined hepatocellular-cholangiocarcinoma (CHC), a histopathologic intermediate form between HCC and CC, suggests phenotypic overlap between these tumors. By applying an integrative oncogenomic approach, we identified a novel HCC subtype, i.e., cholangiocarcinoma-like HCC (CLHCC), which expressed cholangiocarcinoma-like traits (CC signature). In addition, we found that CLHCC coexpressed embryonic stem cell–like expression traits (ES signature) suggesting its derivation from bipotent hepatic progenitor cells. Further histopathological evaluation revealed a variant HCC with fibrous stromal component, i.e. scirrhous HCC, has CC-like genomic features, suggesting that the fibrous stromal component in HCC may contribute to the acquisition of CC-like gene expression trait in HCC. Our integrative analysis combining histopathological and genomic data would be a powerful approach to delineate the tumor heterogeneity.


 E-submission
E-submission THE KOREAN LIVER CANCER ASSOCIATION
THE KOREAN LIVER CANCER ASSOCIATION
 First
First Prev
Prev



 Follow JLC on Twitter
Follow JLC on Twitter